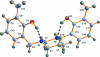
6,6'-Dimethyl-2,2'-[1,3-diazinane-1,3-diyl-bis(methyl-ene)]diphenol
- PMID: 22412589
- PMCID: PMC3295478
- DOI: 10.1107/S1600536812005284
6,6'-Dimethyl-2,2'-[1,3-diazinane-1,3-diyl-bis(methyl-ene)]diphenol
Abstract
In the mol-ecule of the title compound, C(20)H(26)N(2)O(2), the 1,3-diazinane ring adopts a slightly distorted chair conformation and the hy-droxy-benzyl substituents occupy equatorial positions on the N atoms of the heterocyclic ring. There are two intra-molecular O-H⋯N hydrogen bonds between the N atoms of the 1,3-diazinane ring and the hy-droxy groups of the aromatic rings, with an S(6) set-graph motif. However, the two observed intra-molecular hydrogen-bond distances were different. Considering that both N atoms experience the same chemical environment, it is surprising to see the difference in O⋯N distances [2.6771 (14) and 2.8123 (12) Å]. The crystal structure is further stabilized by a C-H⋯π interaction.
Figures
References
-
- Agilent (2010). CrysAlis PRO Agilent Technologies, Yarnton, Oxfordshire, England.
-
- Allen, F. H., Kennard, O., Watson, D. G., Brammer, L., Orpen, A. G. & Taylor, R. (1987). J. Chem. Soc. Perkin Trans. 2, pp. S1–19.
-
- Bernstein, J., Davis, R. E., Shimoni, L. & Chang, N.-L. (1995). Angew. Chem. Int. Ed. Engl. 34, 1555–1573.
-
- Brandenburg, K. & Putz, H. (2005). DIAMOND Crystal Impact, Bonn, Germany.
-
- Burla, M. C., Camalli, M., Carrozzini, B., Cascarano, G. L., Giacovazzo, C., Polidori, G. & Spagna, R. (2003). J. Appl. Cryst. 36, 1103.
LinkOut - more resources
Full Text Sources
Miscellaneous