Impaired myogenic constriction of the renal afferent arteriole in a mouse model of reduced βENaC expression
- PMID: 22419697
- PMCID: PMC3378172
- DOI: 10.1152/ajprenal.00638.2011
Impaired myogenic constriction of the renal afferent arteriole in a mouse model of reduced βENaC expression
Abstract
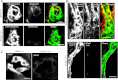
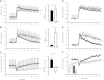
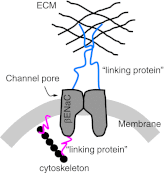
Previous studies demonstrate a role for β epithelial Na(+) channel (βENaC) protein as a mediator of myogenic constriction in renal interlobar arteries. However, the importance of βENaC as a mediator of myogenic constriction in renal afferent arterioles, the primary site of development of renal vascular resistance, has not been determined. We colocalized βENaC with smooth muscle α-actin in vascular smooth muscle cells in renal arterioles using immunofluorescence. To determine the importance of βENaC in myogenic constriction in renal afferent arterioles, we used a mouse model of reduced βENaC (βENaC m/m) and examined pressure-induced constrictor responses in the isolated afferent arteriole-attached glomerulus preparation. We found that, in response to a step increase in perfusion pressure from 60 to 120 mmHg, the myogenic tone increased from 4.5 ± 3.7 to 27.3 ± 5.2% in +/+ mice. In contrast, myogenic tone failed to increase with the pressure step in m/m mice (3.9 ± 0.8 to 6.9 ± 1.4%). To determine the importance of βENaC in myogenic renal blood flow (RBF) regulation, we examined the rate of change in renal vascular resistance following a step increase in perfusion pressure in volume-expanded animals. We found that, following a step increase in pressure, the rate of myogenic correction of RBF is inhibited by 75% in βENaC m/m mice. These findings demonstrate that myogenic constriction in afferent arterioles is dependent on normal expression of βENaC.
Figures

Similar articles
-
βENaC and ASIC2 associate in VSMCs to mediate pressure-induced constriction in the renal afferent arteriole.Am J Physiol Renal Physiol. 2022 May 1;322(5):F498-F511. doi: 10.1152/ajprenal.00003.2022. Epub 2022 Mar 14. Am J Physiol Renal Physiol. 2022. PMID: 35285274 Free PMC article.
-
βENaC is required for whole cell mechanically gated currents in renal vascular smooth muscle cells.Am J Physiol Renal Physiol. 2013 Jun 15;304(12):F1428-37. doi: 10.1152/ajprenal.00444.2012. Epub 2013 Apr 3. Am J Physiol Renal Physiol. 2013. PMID: 23552864 Free PMC article.
-
Altered whole kidney blood flow autoregulation in a mouse model of reduced beta-ENaC.Am J Physiol Renal Physiol. 2010 Feb;298(2):F285-92. doi: 10.1152/ajprenal.00496.2009. Epub 2009 Nov 4. Am J Physiol Renal Physiol. 2010. PMID: 19889952 Free PMC article.
-
Cellular signalling in arteriolar myogenic constriction: involvement of tyrosine phosphorylation pathways.Clin Exp Pharmacol Physiol. 2002 Jul;29(7):612-9. doi: 10.1046/j.1440-1681.2002.03698.x. Clin Exp Pharmacol Physiol. 2002. PMID: 12060106 Review.
-
Altered myogenic responsiveness of the renal microvasculature in experimental hypertension.J Hypertens. 1996 Dec;14(12):1387-401. doi: 10.1097/00004872-199612000-00002. J Hypertens. 1996. PMID: 8986920 Review.
Cited by
-
Endogenously produced 20-HETE modulates myogenic and TGF response in microperfused afferent arterioles.Prostaglandins Other Lipid Mediat. 2013 Apr-May;102-103:42-8. doi: 10.1016/j.prostaglandins.2013.03.001. Epub 2013 Mar 14. Prostaglandins Other Lipid Mediat. 2013. PMID: 23500064 Free PMC article.
-
Tumor necrosis factor-α impairs cerebral blood flow in pregnant rats: role of vascular β-epithelial Na+ channel.Am J Physiol Heart Circ Physiol. 2020 Apr 1;318(4):H1018-H1027. doi: 10.1152/ajpheart.00744.2019. Epub 2020 Mar 13. Am J Physiol Heart Circ Physiol. 2020. PMID: 32167780 Free PMC article.
-
Enhanced maximal exercise capacity, vasodilation to electrical muscle contraction, and hind limb vascular density in ASIC1a null mice.Physiol Rep. 2017 Aug;5(15):e13368. doi: 10.14814/phy2.13368. Physiol Rep. 2017. PMID: 28784852 Free PMC article.
-
ENaC in Salt-Sensitive Hypertension: Kidney and Beyond.Curr Hypertens Rep. 2020 Aug 27;22(9):69. doi: 10.1007/s11906-020-01067-9. Curr Hypertens Rep. 2020. PMID: 32852643 Free PMC article. Review.
-
Renal autoregulation in health and disease.Physiol Rev. 2015 Apr;95(2):405-511. doi: 10.1152/physrev.00042.2012. Physiol Rev. 2015. PMID: 25834230 Free PMC article. Review.
References
-
- Benos DJ, Awayda MS, Ismailov II, Johnson JP. Structure and function of amiloride-sensitive Na+ channels. J Membr Biol 143: 1–18, 1995 - PubMed
-
- Bianchi L, Driscoll M. Protons at the gate: DEG/ENaC ion channels help us feel and remember. Neuron 34: 337–340, 2002 - PubMed
-
- Cupples WA, Braam B. Assessment of renal autoregulation. Am J Physiol Renal Physiol 292: F1105–F1123, 2007 - PubMed
-
- Cupples WA, Novak P, Novak V, Salevsky FC. Spontaneous blood pressure fluctuations and renal blood flow dynamics. Am J Physiol Renal Fluid Electrolyte Physiol 270: F82–F89, 1996 - PubMed
-
- Davis MJ, Hill MA. Signaling mechanisms underlying the vascular myogenic response. Physiol Rev 79: 387–423, 1999 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

