Postnatal development of chorda tympani axons in the rat nucleus of the solitary tract
- PMID: 22430892
- PMCID: PMC3418376
- DOI: 10.1002/cne.23093
Postnatal development of chorda tympani axons in the rat nucleus of the solitary tract
Abstract
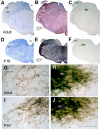
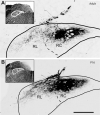
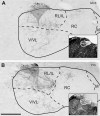
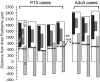
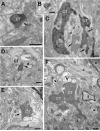
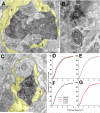
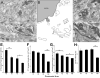
The chorda tympani nerve (CT), one of three nerves that convey gustatory information to the nucleus of the solitary tract (NTS), displays terminal field reorganization after postnatal day 15 in the rat. Aiming to gain insight into mechanisms of this phenomenon, CT axon projection field and terminal morphology in NTS subdivisions were examined using tract tracing, light microscopy, and immunoelectron microscopy at four postnatal ages: P15, P25, P35, and adult. The CT axons that innervated NTS rostrolateral subdivision both in the adult and in P15 rats were morphologically distinct from those that innervated the rostrocentral, gustatory subdivision. In both subdivisions, CT terminals reached morphological maturity before P15. Rostrolateral, but not rostrocentral axons, went through substantial axonal branch elimination after P15. Rostrocentral CT synapses, however, redistribute onto postsynaptic targets in the following weeks. CT terminal preference for GABAergic postsynaptic targets was drastically reduced after P15. Furthermore, CT synapses became a smaller component of the total synaptic input to the rostrocentral NTS after P35. The results underlined that CT axons in rostrocentral and rostrolateral subdivisions represent two distinct populations of CT input, displaying different morphological properties and structural reorganization mechanisms during postnatal development.
Copyright © 2012 Wiley Periodicals, Inc.
Figures


Similar articles
-
A survey of oral cavity afferents to the rat nucleus tractus solitarii.J Comp Neurol. 2012 Feb 15;520(3):495-527. doi: 10.1002/cne.22715. J Comp Neurol. 2012. PMID: 21800298 Free PMC article.
-
Chorda tympani nerve terminal field maturation and maintenance is severely altered following changes to gustatory nerve input to the nucleus of the solitary tract.J Neurosci. 2011 May 25;31(21):7591-603. doi: 10.1523/JNEUROSCI.0151-11.2011. J Neurosci. 2011. PMID: 21613473 Free PMC article.
-
Modifications of gustatory nerve synapses onto nucleus of the solitary tract neurons induced by dietary sodium-restriction during development.J Comp Neurol. 2008 Jun 1;508(4):529-41. doi: 10.1002/cne.21708. J Comp Neurol. 2008. PMID: 18366062 Free PMC article.
-
Impact of chorda tympani nerve injury on cell survival, axon maintenance, and morphology of the chorda tympani nerve terminal field in the nucleus of the solitary tract.J Comp Neurol. 2012 Aug 1;520(11):2395-413. doi: 10.1002/cne.23044. J Comp Neurol. 2012. PMID: 22237830 Free PMC article.
-
Gustatory terminal field organization and developmental plasticity in the nucleus of the solitary tract revealed through triple-fluorescence labeling.J Comp Neurol. 2006 Aug 1;497(4):658-69. doi: 10.1002/cne.21023. J Comp Neurol. 2006. PMID: 16739199 Free PMC article.
Cited by
-
Selective Removal of Sodium Salt Taste Disrupts the Maintenance of Dendritic Architecture of Gustatory Relay Neurons in the Mouse Nucleus of the Solitary Tract.eNeuro. 2020 Oct 28;7(5):ENEURO.0140-20.2020. doi: 10.1523/ENEURO.0140-20.2020. Print 2020 Sep/Oct. eNeuro. 2020. PMID: 32817119 Free PMC article.
-
The μ-opioid receptor agonist DAMGO presynaptically suppresses solitary tract-evoked input to neurons in the rostral solitary nucleus.J Neurophysiol. 2013 Jun;109(11):2815-26. doi: 10.1152/jn.00711.2012. Epub 2013 Mar 13. J Neurophysiol. 2013. PMID: 23486207 Free PMC article.
-
P2X2 Receptor Terminal Field Demarcates a "Transition Zone" for Gustatory and Mechanosensory Processing in the Mouse Nucleus Tractus Solitarius.Chem Senses. 2016 Jul;41(6):515-24. doi: 10.1093/chemse/bjw055. Epub 2016 Apr 30. Chem Senses. 2016. PMID: 27131102 Free PMC article.
-
Transcriptomes and neurotransmitter profiles of classes of gustatory and somatosensory neurons in the geniculate ganglion.Nat Commun. 2017 Oct 2;8(1):760. doi: 10.1038/s41467-017-01095-1. Nat Commun. 2017. PMID: 28970527 Free PMC article.
-
Morphology and connectivity of parabrachial and cortical inputs to gustatory thalamus in rats.J Comp Neurol. 2015 Jan 1;523(1):139-61. doi: 10.1002/cne.23673. Epub 2014 Oct 7. J Comp Neurol. 2015. PMID: 25186035 Free PMC article.
References
-
- Bao H, Bradley RM, Mistretta CM. Development of intrinsic electrophysiological properties in neurons from the gustatory region of rat nucleus of solitary tract. Brain Res Dev Brain Res. 1995;86(1-2):143–154. - PubMed
-
- Bickford ME, Gunluk AE, Van Horn SC, Sherman SM. GABAergic projection from the basal forebrain to the visual sector of the thalamic reticular nucleus in the cat. J Comp Neurol. 1994;348(4):481–510. - PubMed
-
- Biedenbach MA, Chan KY. Tongue mechanoreceptors: comparison of afferent fibers in the lingual nerve and chorda tympani. Brain research. 1971;35(2):584–588. - PubMed
-
- Brown M, Renehan WE, Schweitzer L. Changes in GABA-immunoreactivity during development of the rostral subdivision of the nucleus of the solitary tract. Neuroscience. 2000;100(4):849–859. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

