Dialkylbiaryl Phosphines in Pd-Catalyzed Amination: A User's Guide
- PMID: 22432049
- PMCID: PMC3306613
- DOI: 10.1039/C0SC00331J
Dialkylbiaryl Phosphines in Pd-Catalyzed Amination: A User's Guide
Abstract
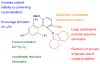
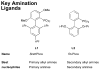
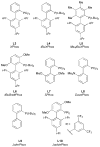
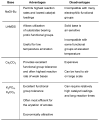
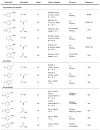
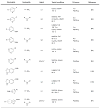
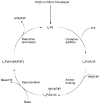
Dialkylbiaryl phosphines are a valuable class of ligand for Pd-catalyzed amination reactions and have been applied in a range of contexts. This review attempts to aid the reader in the selection of the best choice of reaction conditions and ligand of this class for the most commonly encountered and practically important substrate combinations.
Figures


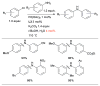

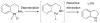
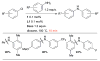
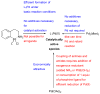
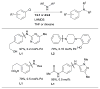
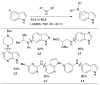
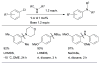
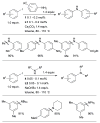
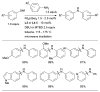
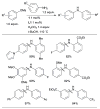
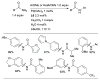
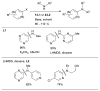
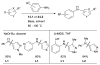
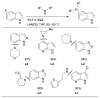
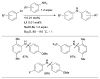
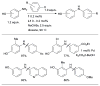
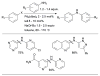
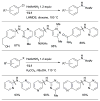
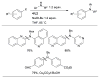

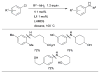


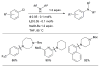
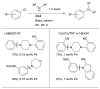
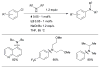
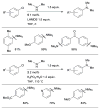
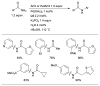
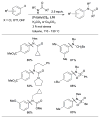
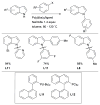
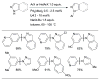

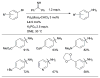
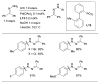
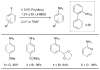
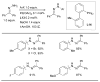
References
-
- King AO, Yasuda N. Topics in Organometallic Chemistry. 2004;6:205–245.
-
- Torborg C, Beller M. Adv Synth Catal. 2009;351:3027–3043.
-
- Carey JS, Laffan D, Thomson C, Williams MT. Org Biomol Chem. 2006;4:2337–2347. - PubMed
-
- Baeza A, Burgos C, Alvarez-Builla J, Vaquero JJ. Tetrahedron Lett. 2007;48:2597–2601.
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

