Regulation of macrophage and dendritic cell responses by their lineage precursors
- PMID: 22433183
- PMCID: PMC3498093
- DOI: 10.1159/000335733
Regulation of macrophage and dendritic cell responses by their lineage precursors
Abstract
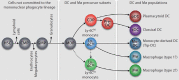
Tissue macrophages (Mø) and dendritic cells (DC) are thought to derive from hematopoietic stem cells, which exist in the bone marrow and generate intermediate precursor populations with increasingly restricted lineage potentials. There exists several precursors committed to the Mø and DC lineages; these cells exhibit distinct tropism and function and respond differentially in pathophysiologic conditions. In this review, we consider experimental contexts in which Mø and DC responses in tissue are not only dictated by the local environment, but also by the quantity and quality of newly recruited lineage precursor cells. Consequently, we discuss whether therapeutic control of Mø and DC responses in tissue may be achieved through manipulation of their lineage precursors.
Copyright © 2012 S. Karger AG, Basel.
Figures

Similar articles
-
Increased generation of dendritic cells from myeloid progenitors in autoimmune-prone nonobese diabetic mice.J Immunol. 2002 May 15;168(10):5032-41. doi: 10.4049/jimmunol.168.10.5032. J Immunol. 2002. PMID: 11994455
-
Developmental stages of myeloid dendritic cells in mouse bone marrow.Int Immunol. 2003 Apr;15(4):515-24. doi: 10.1093/intimm/dxg050. Int Immunol. 2003. PMID: 12663681
-
Lymphoid tissue and plasmacytoid dendritic cells and macrophages do not share a common macrophage-dendritic cell-restricted progenitor.Immunity. 2014 Jul 17;41(1):104-15. doi: 10.1016/j.immuni.2014.05.020. Immunity. 2014. PMID: 25035955
-
Origin, precursors and differentiation of mouse dendritic cells.Nat Rev Immunol. 2003 Jul;3(7):582-90. doi: 10.1038/nri1127. Nat Rev Immunol. 2003. PMID: 12876560 Review.
-
Myb-transformed hematopoietic cells as a model for monocyte differentiation into dendritic cells and macrophages.J Leukoc Biol. 1999 Aug;66(2):217-23. doi: 10.1002/jlb.66.2.217. J Leukoc Biol. 1999. PMID: 10449157 Review.
Cited by
-
The Median Nerve Injury Model in Pre-clinical Research - A Critical Review on Benefits and Limitations.Front Cell Neurosci. 2019 Jun 28;13:288. doi: 10.3389/fncel.2019.00288. eCollection 2019. Front Cell Neurosci. 2019. PMID: 31316355 Free PMC article. Review.
-
Surveillance and countermeasures in innate immunity.J Innate Immun. 2014;6(1):1-2. doi: 10.1159/000357491. J Innate Immun. 2014. PMID: 24611159 Free PMC article. No abstract available.
-
CIRCOAST: a statistical hypothesis test for cellular colocalization with network structures.Bioinformatics. 2019 Feb 1;35(3):506-514. doi: 10.1093/bioinformatics/bty638. Bioinformatics. 2019. PMID: 30032263 Free PMC article.
-
The Janus face of macrophages in immunity.J Innate Immun. 2014;6(6):713-5. doi: 10.1159/000367718. Epub 2014 Sep 10. J Innate Immun. 2014. PMID: 25322689 Free PMC article. No abstract available.
-
Innate immune functions of macrophages in different tissue environments.J Innate Immun. 2012;4(5-6):409-10. doi: 10.1159/000339280. Epub 2012 Jun 12. J Innate Immun. 2012. PMID: 22699934 Free PMC article. No abstract available.
References
-
- Aderem A, Underhill DM. Mechanisms of phagocytosis in macrophages. Annu Rev Immunol. 1999;17:593–623. - PubMed
-
- Janeway CAJ, Medzhitov R. Innate immune recognition. Annu Rev Immunol. 2002;20:197–216. - PubMed
-
- Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. - PubMed
-
- Hansson GK, Libby P. The immune response in atherosclerosis: a double-edged sword. Nat Rev Immunol. 2006;6:508–519. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

