Regulation of GABA and glutamate release from proopiomelanocortin neuron terminals in intact hypothalamic networks
- PMID: 22442070
- PMCID: PMC3321840
- DOI: 10.1523/JNEUROSCI.6032-11.2012
Regulation of GABA and glutamate release from proopiomelanocortin neuron terminals in intact hypothalamic networks
Abstract
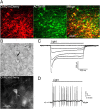
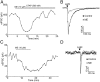
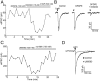
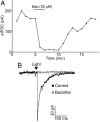
Hypothalamic proopiomelanocortin (POMC) neurons and their peptide products mediate important aspects of energy balance, analgesia, and reward. In addition to peptide products, there is evidence that POMC neurons can also express the amino acid transmitters GABA and glutamate, suggesting these neurons may acutely inhibit or activate downstream neurons. However, the release of amino acid transmitters from POMC neurons has not been thoroughly investigated in an intact system. In the present study, the light-activated cation channel channelrhodopsin-2 (ChR2) was used to selectively evoke transmitter release from POMC neurons. Whole-cell electrophysiologic recordings were made in brain slices taken from POMC-Cre transgenic mice that had been injected with a viral vector containing a floxed ChR2 sequence. Brief pulses of blue light depolarized POMC-ChR2 neurons and induced the release of GABA and glutamate onto unidentified neurons within the arcuate nucleus, as well as onto other POMC neurons. To determine whether the release of GABA and glutamate from POMC terminals can be readily modulated, opioid and GABA(B) receptor agonists were applied. Agonists for μ- and κ-, but not δ-opioid receptors inhibited transmitter release from POMC neurons, as did the GABA(B) receptor agonist baclofen. This regulation indicates that opioids and GABA released from POMC neurons may act at presynaptic receptors on POMC terminals in an autoregulatory manner to limit continued transmission. The results show that, in addition to the relatively slow and long-lasting actions of peptides, POMC neurons can rapidly affect the activity of downstream neurons via GABA and glutamate release.
Figures

Similar articles
-
Differential expression and sensitivity of presynaptic and postsynaptic opioid receptors regulating hypothalamic proopiomelanocortin neurons.J Neurosci. 2011 Jan 5;31(1):281-8. doi: 10.1523/JNEUROSCI.4654-10.2011. J Neurosci. 2011. PMID: 21209213 Free PMC article.
-
GABA release from proopiomelanocortin neurons.J Neurosci. 2004 Feb 18;24(7):1578-83. doi: 10.1523/JNEUROSCI.3952-03.2004. J Neurosci. 2004. PMID: 14973227 Free PMC article.
-
Proopiomelanocortin expression in both GABA and glutamate neurons.J Neurosci. 2009 Oct 28;29(43):13684-90. doi: 10.1523/JNEUROSCI.3770-09.2009. J Neurosci. 2009. PMID: 19864580 Free PMC article.
-
The Relevance of AgRP Neuron-Derived GABA Inputs to POMC Neurons Differs for Spontaneous and Evoked Release.J Neurosci. 2017 Aug 2;37(31):7362-7372. doi: 10.1523/JNEUROSCI.0647-17.2017. Epub 2017 Jun 30. J Neurosci. 2017. PMID: 28667175 Free PMC article.
-
Multiple inhibitory G-protein-coupled receptors resist acute desensitization in the presynaptic but not postsynaptic compartments of neurons.J Neurosci. 2012 Jul 25;32(30):10192-200. doi: 10.1523/JNEUROSCI.1227-12.2012. J Neurosci. 2012. PMID: 22836254 Free PMC article.
Cited by
-
mTORC1 and CB1 receptor signaling regulate excitatory glutamatergic inputs onto the hypothalamic paraventricular nucleus in response to energy availability.Mol Metab. 2019 Oct;28:151-159. doi: 10.1016/j.molmet.2019.08.005. Epub 2019 Aug 8. Mol Metab. 2019. PMID: 31420305 Free PMC article.
-
Glutamate and GABA as rapid effectors of hypothalamic "peptidergic" neurons.Front Behav Neurosci. 2012 Nov 26;6:81. doi: 10.3389/fnbeh.2012.00081. eCollection 2012. Front Behav Neurosci. 2012. PMID: 23189047 Free PMC article.
-
Disruption of GABA or glutamate release from POMC neurons in the adult mouse does not affect metabolic end points.Am J Physiol Regul Integr Comp Physiol. 2020 Nov 1;319(5):R592-R601. doi: 10.1152/ajpregu.00180.2020. Epub 2020 Sep 16. Am J Physiol Regul Integr Comp Physiol. 2020. PMID: 32936679 Free PMC article.
-
GABA type B receptor signaling in proopiomelanocortin neurons protects against obesity, insulin resistance, and hypothalamic inflammation in male mice on a high-fat diet.J Neurosci. 2013 Oct 23;33(43):17166-73. doi: 10.1523/JNEUROSCI.0897-13.2013. J Neurosci. 2013. PMID: 24155320 Free PMC article.
-
Melanocortin 4 receptor is not required for estrogenic regulations on energy homeostasis and reproduction.Metabolism. 2017 May;70:152-159. doi: 10.1016/j.metabol.2016.12.004. Epub 2016 Dec 13. Metabolism. 2017. PMID: 28403939 Free PMC article.
References
-
- Bertolini A, Tacchi R, Vergoni AV. Brain effects of melanocortins. Pharmacol Res. 2009;59:13–47. - PubMed
-
- Chen YY, Pelletier G. Demonstration of contacts between proopiomelanocortin neurons in the rat hypothalamus. Neurosci Lett. 1983;43:271–276. - PubMed
-
- Choudhury AI, Heffron H, Smith MA, Al-Qassab H, Xu AW, Selman C, Simmgen M, Clements M, Claret M, Maccoll G, Bedford DC, Hisadome K, Diakonov I, Moosajee V, Bell JD, Speakman JR, Batterham RL, Barsh GS, Ashford ML, Withers DJ. The role of insulin receptor substrate 2 in hypothalamic and beta cell function. J Clin Invest. 2005;115:940–950. - PMC - PubMed
-
- Coll AP. Effects of pro-opiomelanocortin (POMC) on food intake and body weight: mechanisms and therapeutic potential? Clin Sci (Lond) 2007;113:171–182. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
