Engulfment protein GULP is regulator of transforming growth factor-β response in ovarian cells
- PMID: 22451657
- PMCID: PMC3370247
- DOI: 10.1074/jbc.M111.314997
Engulfment protein GULP is regulator of transforming growth factor-β response in ovarian cells
Abstract
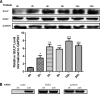
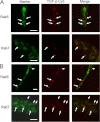
Transforming growth factor β (TGF-β) is a key regulatory molecule with pleiotropic effects on cell growth, migration, and invasion. As a result, impairment of proper TGF-β signaling is central to tumorigenesis and metastasis. The TGF-β receptor V (TGFBRV or LRP1) has been shown to be responsible for TGF-β-mediated cell growth inhibition in Chinese hamster ovary (CHO) cells. The LRP1 adapter protein GULP mediates internalization of the various LRP1-specific ligands, and we hypothesize that GULP acts as a novel regulator of TGF-β signaling in ovarian cells. CHO cells that overexpress exogenous GULP (FL) demonstrate enhancement in growth inhibition, migration, and invasion from TGF-β treatment, whereas cells that lack GULP (AS) show impairment of growth inhibition and decreased migration and invasion. The enhanced TGF-β response in FL cells was confirmed by a prolonged TGF-β-induced SMAD3 phosphorylation, whereas a shortening of the phosphorylation event is observed in AS cells. Mechanistically, the presence of GULP retains the TGF-β in a signaling-competent early endosome for enhanced signaling. To address this mechanism in a physiological setting, TGF-β insensitive ovarian adenocarcinoma cells (HEY) have a very low GULP expression level, similar to the observation made in a wide selection of human ovarian adenocarcinomas. Transfection of GULP into the HEY cells restored the TGF-β responsiveness, as measured by SMAD3 phosphorylation and impairment of cell growth. Because GULP expression positively regulates TGF-β signaling leading to growth inhibition, this may represent an attractive target to achieve TGF-β responsiveness in ovarian cells.
Figures








References
-
- Roberts A. B., Sporn M. B. (1985) Transforming growth factors. Cancer Surv. 4, 683–705 - PubMed
-
- Sporn M. B., Roberts A. B., Wakefield L. M., Assoian R. K. (1986) Transforming growth factor-β. Biological function and chemical structure. Science 233, 532–534 - PubMed
-
- Zhou L., Leung B. S. (1992) Growth regulation of ovarian cancer cells by epidermal growth factor and transforming growth factors α and β1. Biochim. Biophys. Acta 1180, 130–136 - PubMed
-
- Massagué J. (1998) TGF-β signal transduction. Annu. Rev. Biochem. 67, 753–791 - PubMed
-
- Blobe G. C., Schiemann W. P., Lodish H. F. (2000) Role of transforming growth factor β in human disease. N. Engl. J. Med. 342, 1350–1358 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

