Clinical evaluation of a microwave device for treating axillary hyperhidrosis
- PMID: 22452511
- PMCID: PMC3489040
- DOI: 10.1111/j.1524-4725.2012.02375.x
Clinical evaluation of a microwave device for treating axillary hyperhidrosis
Abstract
Background: A third-generation microwave-based device has been developed to treat axillary hyperhidrosis by selectively heating the interface between the skin and underlying fat where the sweat glands reside.
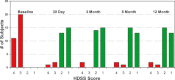
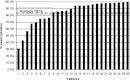
Materials and methods: Thirty-one (31) adults with primary axillary hyperhidrosis were enrolled. All subjects had one to three procedure sessions over a 6-month period to treat both axillae fully. Efficacy was assessed using the Hyperhidrosis Disease Severity Scale (HDSS), gravimetric weight of sweat, and the Dermatologic Life Quality Index (DLQI), a dermatology-specific quality-of-life scale. Subject safety was assessed at each visit. Subjects were followed for 12 months after all procedure sessions were complete.
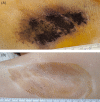
Results: At the 12-month follow-up visit, 90.3% had HDSS scores of 1 or 2, 90.3% had at least a 50% reduction in axillary sweat from baseline, and 85.2% had a reduction of at least 5 points on the DLQI. All subjects experienced transient effects in the treatment area such as swelling, discomfort, and numbness. The most common adverse event (12 subjects) was the presence of altered sensation in the skin of the arm that resolved in all subjects.
Conclusion: The device tested provided efficacious and durable treatment for axillary hyperhidrosis.
© 2012 by the American Society for Dermatologic Surgery, Inc. Published by Wiley Periodicals, Inc.
Figures

Similar articles
-
The efficacy of a microwave device for treating axillary hyperhidrosis and osmidrosis in Asians: a preliminary study.J Cosmet Laser Ther. 2013 Oct;15(5):255-9. doi: 10.3109/14764172.2013.807114. Epub 2013 Jun 21. J Cosmet Laser Ther. 2013. PMID: 23713761
-
A randomized, blinded clinical evaluation of a novel microwave device for treating axillary hyperhidrosis: the dermatologic reduction in underarm perspiration study.Dermatol Surg. 2012 Feb;38(2):185-91. doi: 10.1111/j.1524-4725.2011.02250.x. Epub 2012 Jan 30. Dermatol Surg. 2012. PMID: 22289389 Clinical Trial.
-
A survey of long-term results with microwave energy device for treating axillary hyperhidrosis.J Cosmet Laser Ther. 2021 May 19;23(3-4):49-51. doi: 10.1080/14764172.2021.1957115. Epub 2021 Aug 19. J Cosmet Laser Ther. 2021. PMID: 34409892
-
Treatment of hyperhidrosis with microwave technology.Semin Cutan Med Surg. 2013 Mar;32(1):2-8. Semin Cutan Med Surg. 2013. PMID: 24049923 Review.
-
Microwave Treatment for Axillary Hyperhidrosis and Bromhidrosis.Actas Dermosifiliogr. 2017 Jun;108(5):418-422. doi: 10.1016/j.ad.2016.12.011. Epub 2017 Mar 9. Actas Dermosifiliogr. 2017. PMID: 28284421 Review. English, Spanish.
Cited by
-
Conservative Management of Median Nerve Brachial Plexopathy after Microwave-based MiraDry Treatment for Axillary Hyperhidrosis.Plast Reconstr Surg Glob Open. 2021 Dec 15;9(12):e3992. doi: 10.1097/GOX.0000000000003992. eCollection 2021 Dec. Plast Reconstr Surg Glob Open. 2021. PMID: 34926103 Free PMC article.
-
Botulinum toxin A versus microwave thermolysis for primary axillary hyperhidrosis: A randomized controlled trial.JAAD Int. 2024 Jan 23;15:91-99. doi: 10.1016/j.jdin.2023.12.011. eCollection 2024 Jun. JAAD Int. 2024. PMID: 38495540 Free PMC article.
-
Management of hyperhidrosis.Clin Cosmet Investig Dermatol. 2014 Oct 29;7:285-99. doi: 10.2147/CCID.S53119. eCollection 2014. Clin Cosmet Investig Dermatol. 2014. PMID: 25378942 Free PMC article. Review.
-
Evaluation of Efficacy and Safety of miraDry® Procedure in the Treatment of Primary Axillary Hyperhidrosis.Aesthetic Plast Surg. 2025 May;49(9):2545-2551. doi: 10.1007/s00266-024-04558-1. Epub 2025 Jan 3. Aesthetic Plast Surg. 2025. PMID: 39753871
-
Hyperhidrosis: an update on prevalence and severity in the United States.Arch Dermatol Res. 2016 Dec;308(10):743-749. doi: 10.1007/s00403-016-1697-9. Epub 2016 Oct 15. Arch Dermatol Res. 2016. PMID: 27744497 Free PMC article.
References
-
- Strutton DR, Kowalski JW, Glaser DA, Stang PE. US prevalence of hyperhidrosis and impact on individuals with axillary hyperhidrosis: results from national survey. J Am Acad Dermatol. 2004;51:241–8. - PubMed
-
- Solish N, Bertucci V, Dansereau A, Hong HC-H, et al. A comprehensive approach to the recognition, diagnosis, and severity-based treatment of focal hyperhidrosis: recommendations of the Canadian Hyperhidrosis Advisory Committee. Dermatol Surg. 2007;33:908–23. - PubMed
-
- Johnson JE, O'Shaughnessy KF, Kim S. Microwave thermolysis of sweat glands. Lasers Surg Med. 2012;44:20–5. - PubMed
-
- Glaser DA, Coleman WP, Fan LK, Kaminer MS, et al. A randomized, blinded clinical evaluation of a novel microwave device for treating axillary hyperhidrosis: the Dermatologic Reduction In Underarm Perspiration Study. Dermatol Surg. 2012;38:185–91. - PubMed
-
- Kowalski JW, Eadie N, Dagget S, Lai P-Y. Validity and reliability of the Hyperhidrosis Disease Severity Scale (HDSS) J Am Acad Dermatol. 2004;36:P51. Poster presented at the 62nd Annual Meeting of the American Academy of Dermatology; February 6–10, 2004; Washington DC. Poster P198.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

