Peroxisome deficiency but not the defect in ether lipid synthesis causes activation of the innate immune system and axonal loss in the central nervous system
- PMID: 22458306
- PMCID: PMC3419640
- DOI: 10.1186/1742-2094-9-61
Peroxisome deficiency but not the defect in ether lipid synthesis causes activation of the innate immune system and axonal loss in the central nervous system
Abstract
Background: Mice with peroxisome deficiency in neural cells (Nestin-Pex5-/-) develop a neurodegenerative phenotype leading to motor and cognitive disabilities and early death. Major pathologies at the end stage of disease include severe demyelination, axonal degeneration and neuroinflammation. We now investigated the onset and progression of these pathological processes, and their potential interrelationship. In addition, the putative role of oxidative stress, the impact of plasmalogen depletion on the neurodegenerative phenotype, and the consequences of peroxisome elimination in the postnatal period were studied.
Methods: Immunohistochemistry in association with gene expression analysis was performed on Nestin-Pex5-/- mice to document demyelination, axonal damage and neuroinflammation. Also Gnpat-/- mice, with selective plasmalogen deficiency and CMV-Tx-Pex5-/- mice, with tamoxifen induced generalized loss of peroxisomes were analysed.
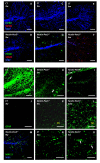
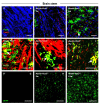
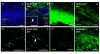
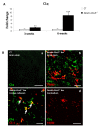
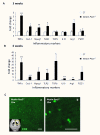
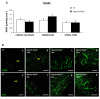
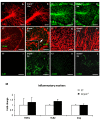
Results: Activation of the innate immune system is a very early event in the pathological process in Nestin-Pex5-/- mice which evolves in chronic neuroinflammation. The complement factor C1q, one of the earliest up regulated transcripts, was expressed on neurons and oligodendrocytes but not on microglia. Transcripts of other pro- and anti-inflammatory genes and markers of phagocytotic activity were already significantly induced before detecting pathologies with immunofluorescent staining. Demyelination, macrophage activity and axonal loss co-occurred throughout the brain. As in patients with mild peroxisome biogenesis disorders who develop regressive changes, demyelination in cerebellum and brain stem preceded major myelin loss in corpus callosum of both Nestin-Pex5-/- and CMV-Tx-Pex5-/- mice. These lesions were not accompanied by generalized oxidative stress throughout the brain. Although Gnpat-/- mice displayed dysmyelination and Purkinje cell axon damage in cerebellum, confirming previous observations, no signs of inflammation or demyelination aggravating with age were observed.
Conclusions: Peroxisome inactivity triggers a fast neuroinflammatory reaction, which is not solely due to the depletion of plasmalogens. In association with myelin abnormalities this causes axon damage and loss.
Figures


Similar articles
-
Absence of functional peroxisomes from mouse CNS causes dysmyelination and axon degeneration.J Neurosci. 2008 Apr 9;28(15):4015-27. doi: 10.1523/JNEUROSCI.4968-07.2008. J Neurosci. 2008. PMID: 18400901 Free PMC article.
-
Axonal integrity in the absence of functional peroxisomes from projection neurons and astrocytes.Glia. 2010 Oct;58(13):1532-43. doi: 10.1002/glia.21027. Glia. 2010. PMID: 20578053
-
Leukodystrophy caused by plasmalogen deficiency rescued by glyceryl 1-myristyl ether treatment.Brain Pathol. 2019 Sep;29(5):622-639. doi: 10.1111/bpa.12710. Epub 2019 Feb 20. Brain Pathol. 2019. PMID: 30667116 Free PMC article.
-
Peroxisomes, myelination, and axonal integrity in the CNS.Neuroscientist. 2009 Aug;15(4):367-79. doi: 10.1177/1073858409336297. Neuroscientist. 2009. PMID: 19666893 Review.
-
Oligodendroglial impact on axonal function and survival - a hypothesis.Curr Opin Neurol. 2008 Jun;21(3):235-41. doi: 10.1097/WCO.0b013e328300c71f. Curr Opin Neurol. 2008. PMID: 18451704 Review.
Cited by
-
Miro-Working beyond Mitochondria and Microtubules.Cells. 2018 Mar 4;7(3):18. doi: 10.3390/cells7030018. Cells. 2018. PMID: 29510535 Free PMC article.
-
Peroxisome Deficiency in Cochlear Hair Cells Causes Hearing Loss by Deregulating BK Channels.Adv Sci (Weinh). 2023 Jul;10(20):e2300402. doi: 10.1002/advs.202300402. Epub 2023 May 12. Adv Sci (Weinh). 2023. PMID: 37171794 Free PMC article.
-
Neuronal Dysfunction and Behavioral Abnormalities Are Evoked by Neural Cells and Aggravated by Inflammatory Microglia in Peroxisomal β-Oxidation Deficiency.Front Cell Neurosci. 2018 May 23;12:136. doi: 10.3389/fncel.2018.00136. eCollection 2018. Front Cell Neurosci. 2018. PMID: 29892213 Free PMC article.
-
Functional multi-organelle units control inflammatory lipid metabolism of macrophages.Nat Cell Biol. 2024 Aug;26(8):1261-1273. doi: 10.1038/s41556-024-01457-0. Epub 2024 Jul 5. Nat Cell Biol. 2024. PMID: 38969763 Free PMC article.
-
Role and Function of Peroxisomes in Neuroinflammation.Cells. 2024 Oct 5;13(19):1655. doi: 10.3390/cells13191655. Cells. 2024. PMID: 39404418 Free PMC article. Review.
References
-
- Hulshagen L, Krysko O, Huyghe S, Klein R, Van Veldhoven PP, De Deyn PP, D’hooge R, Hartmann D, Baes M. Absence of functional peroxisomes from mouse central nervous system causes dysmyelination and axon degeneration. J Neurosci. 2008;28:4015–4027. doi: 10.1523/JNEUROSCI.4968-07.2008. - DOI - PMC - PubMed
-
- Krysko O, Hulshagen L, Janssen A, Schutz G, Klein R, De Bruycker M, Espeel M, Gressens P, Baes M. Neocortical and cerebellar developmental abnormalities in conditions of selective elimination of peroxisomes from brain or from liver. J Neurosci Res. 2007;85:58–72. doi: 10.1002/jnr.21097. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

