Effect of chronic L-dopa or melatonin treatments after dopamine deafferentation in rats: dyskinesia, motor performance, and cytological analysis
- PMID: 22462019
- PMCID: PMC3302121
- DOI: 10.5402/2012/360379
Effect of chronic L-dopa or melatonin treatments after dopamine deafferentation in rats: dyskinesia, motor performance, and cytological analysis
Abstract
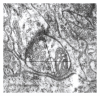
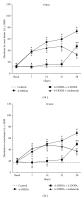
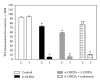
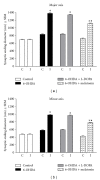
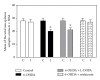
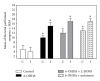
The present study examines the ability of melatonin to protect striatal dopaminergic loss induced by 6-OHDA in a rat model of Parkinson's disease, comparing the results with L-DOPA-treated rats. The drugs were administered orally daily for a month, their therapeutic or dyskinetic effects were assessed by means of abnormal involuntary movements (AIMs) and stepping ability. At the cellular level, the response was evaluated using tyrosine hydroxylase immunoreactivity and striatal ultrastructural changes to compare between L-DOPA-induced AIMs and Melatonin-treated rats. Our findings demonstrated that chronic oral administration of Melatonin improved the alterations caused by the neurotoxin 6-OHDA. Melatonin-treated animals perform better in the motor tasks and had no dyskinetic alterations compared to L-DOPA-treated group. At the cellular level, we found that Melatonin-treated rats showed more TH-positive neurons and their striatal ultrastructure was well preserved. Thus, Melatonin is a useful treatment to delay the cellular and behavioral alterations observed in Parkinson's disease.
Figures


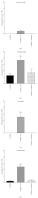

Similar articles
-
5-Hydroxy-tryptophan for the treatment of L-DOPA-induced dyskinesia in the rat Parkinson's disease model.Neurobiol Dis. 2013 Dec;60:108-14. doi: 10.1016/j.nbd.2013.08.014. Epub 2013 Sep 1. Neurobiol Dis. 2013. PMID: 24004632
-
Differential induction of dyskinesia and neuroinflammation by pulsatile versus continuous l-DOPA delivery in the 6-OHDA model of Parkinson's disease.Exp Neurol. 2016 Dec;286:83-92. doi: 10.1016/j.expneurol.2016.09.013. Epub 2016 Sep 30. Exp Neurol. 2016. PMID: 27697481
-
Anti-dyskinetic effect of anpirtoline in animal models of L-DOPA-induced dyskinesia.Neurosci Res. 2013 Dec;77(4):242-6. doi: 10.1016/j.neures.2013.10.002. Epub 2013 Oct 14. Neurosci Res. 2013. PMID: 24135129
-
Melatonin enhances L-DOPA therapeutic effects, helps to reduce its dose, and protects dopaminergic neurons in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced Parkinsonism in mice.J Pineal Res. 2015 Apr;58(3):262-74. doi: 10.1111/jpi.12212. Epub 2015 Feb 23. J Pineal Res. 2015. PMID: 25626558
-
Animal models of L-DOPA-induced dyskinesia: the 6-OHDA-lesioned rat and mouse.J Neural Transm (Vienna). 2018 Aug;125(8):1137-1144. doi: 10.1007/s00702-017-1825-5. Epub 2017 Dec 14. J Neural Transm (Vienna). 2018. PMID: 29242978 Review.
Cited by
-
Melatonin as a Chronobiotic and Cytoprotective Agent in Parkinson's Disease.Front Pharmacol. 2021 Apr 15;12:650597. doi: 10.3389/fphar.2021.650597. eCollection 2021. Front Pharmacol. 2021. PMID: 33935759 Free PMC article. Review.
-
Effects of Melatonin Levels on Neurotoxicity of the Medial Prefrontal Cortex in a Rat Model of Parkinson's Disease.Chin Med J (Engl). 2017 Nov 20;130(22):2726-2731. doi: 10.4103/0366-6999.218025. Chin Med J (Engl). 2017. PMID: 29133763 Free PMC article.
-
Targeting neuroendocrine abnormalities in Parkinson's disease with exercise.Front Neurosci. 2023 Sep 8;17:1228444. doi: 10.3389/fnins.2023.1228444. eCollection 2023. Front Neurosci. 2023. PMID: 37746149 Free PMC article.
-
Melatonin for Sleep Disorders in Patients with Neurodegenerative Diseases.Curr Neurol Neurosci Rep. 2016 Jul;16(7):63. doi: 10.1007/s11910-016-0664-3. Curr Neurol Neurosci Rep. 2016. PMID: 27180068 Review.
-
Rapid Eye Movement Sleep Homeostatic Response: A Potential Marker for Early Detection of Parkinson's Disease.J Alzheimers Dis Parkinsonism. 2016 Aug;6(4):255. doi: 10.4172/2161-0460.1000255. Epub 2016 Aug 18. J Alzheimers Dis Parkinsonism. 2016. PMID: 27713856 Free PMC article. No abstract available.
References
-
- Perry TL, Yong VW. Idiopathic Parkinson’s disease, progressive supranuclear palsy and glutathione metabolism in the substantia nigra of patients. Neuroscience Letters . 1986;67(3):269–274. . - PubMed
-
- Saggu H, Cooksey J, Dexter D, et al. A selective increase in particulate superoxide dismutase activity in parkinsonian substantia nigra. Journal of Neurochemistry . 1989;53(3):692–697. . - PubMed
-
- Dexter DT, Wells FR, Lees AJ, et al. Increased nigral iron content and alterations in other metal ions occurring in brain in Parkinson’s disease. Journal of Neurochemistry . 1989;52(6):1830–1836. . - PubMed
-
- Alam ZI, Daniel SE, Lees AJ, Marsden DC, Jenner P, Halliwell B. A generalised increase in protein carbonyls in the brain in Parkinson’s but not incidental Lewy body disease. Journal of Neurochemistry . 1997;69(3):1326–1329. . - PubMed
LinkOut - more resources
Full Text Sources

