The essential role of protein kinase Cδ in diabetes-induced neural tube defects
- PMID: 22463764
- PMCID: PMC4455937
- DOI: 10.3109/14767058.2012.677963
The essential role of protein kinase Cδ in diabetes-induced neural tube defects
Abstract
Background: Maternal diabetes causes neural tube defects (NTDs) in the embryos via activating protein kinase Cs (PKCs), which regulate programmed cell death (apoptosis). The aims of this study are to investigate the role of proapoptotic PKCδ in NTD formation and the underlying mechanisms.
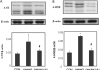
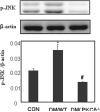
Methods: PKCδ heterozygous (pkcδ(+/-)) female mice were diabetic (DM) induced by intravenous injection of streptozotocin. Occurrence of NTDs was evaluated at embryonic day 11.5 and compared between wild type (WT) and PKCδ homozygous (pkcδ(-/-)) embryos. Changes in oxidative and endoplasmic reticulum (ER) stress-associated factors and stress-response c-Jun N-terminal kinases (JNKs) were assessed using Western blot assay.
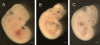
Results: Compared to DM/WT, the DM/PKCδ(-/-) embryos had significantly lower NTD rate and lower levels of oxidative and ER stress factors and JNK activation. These values were similar to those in the non-diabetic control group.
Conclusion: PKCδ plays a critical role in diabetes-induced NTDs, potentially through increasing oxidative and ER stress and JNK-associated stress-response pathways.
Figures
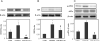
Similar articles
-
Deficiency of the oxidative stress-responsive kinase p70S6K1 restores autophagy and ameliorates neural tube defects in diabetic embryopathy.Am J Obstet Gynecol. 2020 Nov;223(5):753.e1-753.e14. doi: 10.1016/j.ajog.2020.05.015. Epub 2020 May 13. Am J Obstet Gynecol. 2020. PMID: 32416155 Free PMC article.
-
Modulation of nuclear factor-κB signaling and reduction of neural tube defects by quercetin-3-glucoside in embryos of diabetic mice.Am J Obstet Gynecol. 2018 Aug;219(2):197.e1-197.e8. doi: 10.1016/j.ajog.2018.04.045. Epub 2018 May 5. Am J Obstet Gynecol. 2018. PMID: 29733843 Free PMC article.
-
Decoding the oxidative stress hypothesis in diabetic embryopathy through proapoptotic kinase signaling.Am J Obstet Gynecol. 2015 May;212(5):569-79. doi: 10.1016/j.ajog.2014.11.036. Epub 2014 Nov 27. Am J Obstet Gynecol. 2015. PMID: 25434839 Free PMC article. Review.
-
c-Jun NH2-terminal kinase 1/2 and endoplasmic reticulum stress as interdependent and reciprocal causation in diabetic embryopathy.Diabetes. 2013 Feb;62(2):599-608. doi: 10.2337/db12-0026. Epub 2012 Sep 6. Diabetes. 2013. PMID: 22961085 Free PMC article.
-
Recent studies on neural tube defects in embryos of diabetic pregnancy: an overview.Curr Med Chem. 2009;16(18):2345-54. doi: 10.2174/092986709788453069. Curr Med Chem. 2009. PMID: 19519395 Review.
Cited by
-
Tip60- and sirtuin 2-regulated MARCKS acetylation and phosphorylation are required for diabetic embryopathy.Nat Commun. 2019 Jan 17;10(1):282. doi: 10.1038/s41467-018-08268-6. Nat Commun. 2019. PMID: 30655546 Free PMC article.
-
Identification of novel cell survival regulation in diabetic embryopathy via phospholipidomic profiling.Biochem Biophys Res Commun. 2016 Feb 12;470(3):599-605. doi: 10.1016/j.bbrc.2016.01.098. Epub 2016 Jan 19. Biochem Biophys Res Commun. 2016. PMID: 26797275 Free PMC article.
-
Curcumin ameliorates high glucose-induced neural tube defects by suppressing cellular stress and apoptosis.Am J Obstet Gynecol. 2015 Jun;212(6):802.e1-8. doi: 10.1016/j.ajog.2015.01.017. Epub 2015 Jan 13. Am J Obstet Gynecol. 2015. PMID: 25595578 Free PMC article.
-
Genetic, epigenetic, and environmental contributions to neural tube closure.Annu Rev Genet. 2014;48:583-611. doi: 10.1146/annurev-genet-120213-092208. Epub 2014 Oct 6. Annu Rev Genet. 2014. PMID: 25292356 Free PMC article. Review.
-
Exploring Research Trends and Mechanisms: Maternal Diabetes and Neural Tube Defects (1991-2023).J Multidiscip Healthc. 2025 Feb 25;18:1107-1121. doi: 10.2147/JMDH.S501402. eCollection 2025. J Multidiscip Healthc. 2025. PMID: 40026865 Free PMC article.
References
-
- Mills JL. Malformations in infants of diabetic mothers. Teratology. 1982;25:385–394. - PubMed
-
- Reece EA. Obesity, diabetes, and links to congenital defects: a review of the evidence and recommendations for intervention. J Matern Fetal Neonatal Med. 2008;21:173–180. - PubMed
-
- Zhao Z, Reece EA. Experimental mechanisms of diabetic embryopathy and strategies for developing therapeutic interventions. J Soc Gynecol Investig. 2005;12:549–557. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous
