Microarray analysis verifies two distinct phenotypes of glioblastomas resistant to antiangiogenic therapy
- PMID: 22472177
- PMCID: PMC3354006
- DOI: 10.1158/1078-0432.CCR-11-2390
Microarray analysis verifies two distinct phenotypes of glioblastomas resistant to antiangiogenic therapy
Abstract
Purpose: To identify mechanisms and mediators of resistance to antiangiogenic therapy in human glioblastoma.
Experimental design: We carried out microarray gene expression analysis and immunohistochemistry comparing 21 recurrent glioblastomas progressing during antiangiogenic treatment with VEGF neutralizing antibody bevacizumab to paired pretreatment tumors from the same patients.
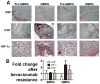
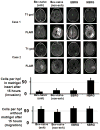
Results: Microarray analysis revealed that bevacizumab-resistant glioblastomas (BRG) had two clustering patterns defining subtypes that reflect radiographic growth patterns. Enhancing BRGs (EBRG) exhibited MRI enhancement, a long-established criterion for glioblastoma progression, and expressed mitogen-activated protein kinases, neural cell adhesion molecule-1 (NCAM-1), and aquaporin 4. Compared with their paired pretreatment tumors, EBRGs had unchanged vascularity and hypoxia, with increased proliferation. Nonenhancing BRGs (NBRG) exhibited minimal MRI enhancement but had FLAIR-bright expansion, a newer criterion for glioblastoma recurrence since the advent of antiangiogenic therapy, and expressed integrin α5, laminin, fibronectin1, and PDGFRβ. NBRGs had less vascularity, more hypoxia, and unchanged proliferation than their paired pretreatment tumors. Primary NBRG cells exhibited more stellate morphology with a 3-fold increased shape factor and were nearly 4-fold more invasive in Matrigel chambers than primary cells from EBRGs or bevacizumab-naive glioblastomas (P < 0.05).
Conclusion: Using microarray analysis, we found two resistance patterns during antiangiogenic therapy with distinct molecular profiles and radiographic growth patterns. These studies provide valuable biologic insight into the resistance that has limited antiangiogenic therapy to date.
©2012 AACR.
Figures



References
-
- Vredenburgh JJ, Desjardins A, Herndon JE, 2nd, Marcello J, Reardon DA, Quinn JA, et al. Bevacizumab plus irinotecan in recurrent glioblastoma multiforme. J Clin Oncol. 2007;25:4722–9. - PubMed
-
- Vredenburgh JJ, Desjardins A, Herndon JE, 2nd, Dowell JM, Reardon DA, Quinn JA, et al. Phase II trial of bevacizumab and irinotecan in recurrent malignant glioma. Clin Cancer Res. 2007;13:1253–9. - PubMed
-
- Friedman HS, Prados MD, Wen PY, Mikkelsen T, Schiff D, Abrey LE, et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol. 2009;27:4733–40. - PubMed
-
- Jain RK, Duda DG, Clark JW, Loeffler JS. Lessons from phase III clinical trials on anti-VEGF therapy for cancer. Nat Clin Pract Oncol. 2006;3:24–40. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

