Podocyte-specific overexpression of human angiotensin-converting enzyme 2 attenuates diabetic nephropathy in mice
- PMID: 22475818
- PMCID: PMC3410252
- DOI: 10.1038/ki.2012.83
Podocyte-specific overexpression of human angiotensin-converting enzyme 2 attenuates diabetic nephropathy in mice
Abstract
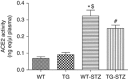
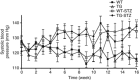
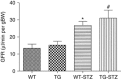
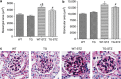
Angiotensin-converting enzyme 2 (ACE2) degrades angiotensin II to angiotensin-(1-7) and is expressed in podocytes. Here we overexpressed ACE2 in podocytes in experimental diabetic nephropathy using transgenic methods where a nephrin promoter drove the expression of human ACE2. Glomeruli from these mice had significantly increased mRNA, protein, and activity of ACE2 compared to wild-type mice. Male mice were treated with streptozotocin to induce diabetes. After 16 weeks, there was no significant difference in plasma glucose levels between wild-type and transgenic diabetic mice. Urinary albumin was significantly increased in wild-type diabetic mice at 4 weeks, whereas albuminuria in transgenic diabetic mice did not differ from wild-type nondiabetic mice. However, this effect was transient and by 16 weeks both transgenic and nontransgenic diabetic mice had similar rates of proteinuria. Compared to wild-type diabetic mice, transgenic diabetic mice had an attenuated increase in mesangial area, decreased glomerular area, and a blunted decrease in nephrin expression. Podocyte numbers decreased in wild-type diabetic mice at 16 weeks, but were unaffected in transgenic diabetic mice. At 8 weeks, kidney cortical expression of transforming growth factor-β1 was significantly inhibited in transgenic diabetic mice as compared to wild-type diabetic mice. Thus, the podocyte-specific overexpression of human ACE2 transiently attenuates the development of diabetic nephropathy.
Figures



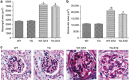
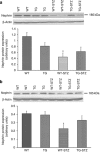
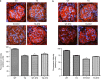
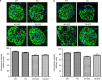

Comment in
-
Podocyte ACE2 protects against diabetic nephropathy.Kidney Int. 2012 Aug;82(3):255-6. doi: 10.1038/ki.2012.127. Kidney Int. 2012. PMID: 22791320 Free PMC article.
References
-
- Donoghue M, Hsieh F, Baronas E, et al. A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1-9. Circ Res. 2000;87:e1–e9. - PubMed
-
- Tipnis S, Hooper N, Hyde R, et al. A human homolog of angiotensin-converting enzyme. J Biol Chem. 2000;275:33238–33243. - PubMed
-
- Freeman EJ, Chisolm GM, Ferrario CM, et al. Angiotensin-(1-7) inhibits vascular smooth muscle cell growth. Hypertension. 1996;28:104–108. - PubMed
-
- Sampaio WO, Henrique de Castro C, Santos RA, et al. Angiotensin-(1-7) counterregulates angiotensin II signaling in human endothelial cells. Hypertension. 2007;50:1093–1098. - PubMed
-
- Su Z, Zimpelmann J, Burns KD. Angiotensin-(1-7) inhibits angiotensin II-stimulated phosphorylation of MAP kinases in proximal tubular cells. Kidney Int. 2006;69:2212–2218. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

