Semaphorin 4D cooperates with VEGF to promote angiogenesis and tumor progression
- PMID: 22476930
- PMCID: PMC3733222
- DOI: 10.1007/s10456-012-9268-y
Semaphorin 4D cooperates with VEGF to promote angiogenesis and tumor progression
Retraction in
-
Retraction Note to: Semaphorin 4D cooperates with VEGF to promote angiogenesis and tumor progression.Angiogenesis. 2020 May;23(2):267. doi: 10.1007/s10456-020-09709-x. Angiogenesis. 2020. PMID: 32157473 Free PMC article.
Abstract
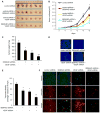
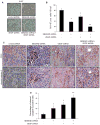
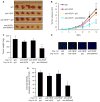
The semaphorins and plexins comprise a family of cysteine-rich proteins implicated in control of nerve growth and development and regulation of the immune response. Our group and others have found that Semaphorin 4D (SEMA4D) and its receptor, Plexin-B1, play an important role in tumor-induced angiogenesis, with some neoplasms producing SEMA4D in a manner analogous to vascular endothelial growth factor (VEGF) in order to attract Plexin-B1-expressing endothelial cells into the tumor for the purpose of promoting growth and vascularity. While anti-VEGF strategies have been the focus of most angiogenesis inhibition research, such treatment can lead to upregulation of pro-angiogenic factors that can compensate for the loss of VEGF, eventually leading to failure of therapy. Here, we demonstrate that SEMA4D cooperates with VEGF to promote angiogenesis in malignancies and can perform the same function in a setting of VEGF blockade. We also show the potential value of inhibiting SEMA4D/Plexin-B1 signaling as a complementary mechanism to anti-VEGF treatment, particularly in VEGF inhibitor-resistant tumors, suggesting that this may represent a novel treatment for some cancers.
Figures
Similar articles
-
The Semaphorin 4D-Plexin-B1-RhoA signaling axis recruits pericytes and regulates vascular permeability through endothelial production of PDGF-B and ANGPTL4.Angiogenesis. 2014 Jan;17(1):261-74. doi: 10.1007/s10456-013-9395-0. Epub 2013 Oct 10. Angiogenesis. 2014. Retraction in: Angiogenesis. 2020 May;23(2):269. doi: 10.1007/s10456-020-09710-4. PMID: 24114199 Free PMC article. Retracted.
-
VEGF and SEMA4D have synergistic effects on the promotion of angiogenesis in epithelial ovarian cancer.Cell Mol Biol Lett. 2018 Jan 3;23:2. doi: 10.1186/s11658-017-0058-9. eCollection 2018. Cell Mol Biol Lett. 2018. PMID: 29308068 Free PMC article.
-
Characterization of the Effects of Semaphorin 4D Signaling on Angiogenesis.Methods Mol Biol. 2017;1493:429-441. doi: 10.1007/978-1-4939-6448-2_31. Methods Mol Biol. 2017. PMID: 27787869 Free PMC article.
-
Roles of Sema4D and Plexin-B1 in tumor progression.Mol Cancer. 2010 Sep 21;9:251. doi: 10.1186/1476-4598-9-251. Mol Cancer. 2010. PMID: 20858260 Free PMC article. Review.
-
Semaphorin signaling in vascular and tumor biology.Adv Exp Med Biol. 2007;600:118-31. doi: 10.1007/978-0-387-70956-7_10. Adv Exp Med Biol. 2007. PMID: 17607951 Review.
Cited by
-
The role of the semaphorins in cancer.Cell Adh Migr. 2016 Nov;10(6):652-674. doi: 10.1080/19336918.2016.1197478. Epub 2016 Aug 17. Cell Adh Migr. 2016. PMID: 27533782 Free PMC article. Review.
-
A Sleeping Beauty forward genetic screen identifies new genes and pathways driving osteosarcoma development and metastasis.Nat Genet. 2015 Jun;47(6):615-24. doi: 10.1038/ng.3293. Epub 2015 May 11. Nat Genet. 2015. PMID: 25961939 Free PMC article.
-
Targeted therapy with apatinib in a patient with relapsed small cell lung cancer: A case report and literature review.Medicine (Baltimore). 2017 Dec;96(50):e9259. doi: 10.1097/MD.0000000000009259. Medicine (Baltimore). 2017. PMID: 29390367 Free PMC article. Review.
-
The Semaphorin 4D-Plexin-B1-RhoA signaling axis recruits pericytes and regulates vascular permeability through endothelial production of PDGF-B and ANGPTL4.Angiogenesis. 2014 Jan;17(1):261-74. doi: 10.1007/s10456-013-9395-0. Epub 2013 Oct 10. Angiogenesis. 2014. Retraction in: Angiogenesis. 2020 May;23(2):269. doi: 10.1007/s10456-020-09710-4. PMID: 24114199 Free PMC article. Retracted.
-
Exposure to PM2.5 via vascular endothelial growth factor relationship: Meta-analysis.PLoS One. 2018 Jun 18;13(6):e0198813. doi: 10.1371/journal.pone.0198813. eCollection 2018. PLoS One. 2018. PMID: 29912914 Free PMC article. Review.
References
-
- Zachary I. Vascular endothelial growth factor and anti-angiogenic peptides as therapeutic and investigational molecules. IDrugs. 2003;6:224–231. - PubMed
-
- Lyden D, Hattori K, Dias S, Costa C, Blaikie P, Butros L, Chadburn A, Heissig B, Marks W, Witte L, Wu Y, Hicklin D, Zhu Z, Hackett NR, Crystal RG, Moore MA, Hajjar KA, Manova K, Benezra R, Rafii S. Impaired recruitment of bone-marrow-derived endothelial and hematopoietic precursor cells blocks tumor angiogenesis and growth. Nat Med. 2001;7:1194–1201. - PubMed
-
- Fischer C, Jonckx B, Mazzone M, Zacchigna S, Loges S, Pattarini L, Chorianopoulos E, Liesenborghs L, Koch M, De Mol M, Autiero M, Wyns S, Plaisance S, Moons L, van Rooijen N, Giacca M, Stassen JM, Dewerchin M, Collen D, Carmeliet P. Anti-PlGF inhibits growth of VEGF(R)-inhibitor-resistant tumors without affecting healthy vessels. Cell. 2007;131:463–475. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

