Protein C anticoagulant and cytoprotective pathways
- PMID: 22477541
- PMCID: PMC3413316
- DOI: 10.1007/s12185-012-1059-0
Protein C anticoagulant and cytoprotective pathways
Abstract
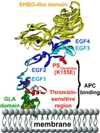
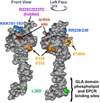
Plasma protein C is a serine protease zymogen that is transformed into the active, trypsin-like protease, activated protein C (APC), which can exert multiple activities. For its anticoagulant action, APC causes inactivation of the procoagulant cofactors, factors Va and VIIIa, by limited proteolysis, and APC's anticoagulant activity is promoted by protein S, various lipids, high-density lipoprotein, and factor V. Hereditary heterozygous deficiency of protein C or protein S is linked to moderately increased risk for venous thrombosis, while a severe or total deficiency of either protein is linked to neonatal purpura fulminans. In recent years, the beneficial direct effects of APC on cells which are mediated by several specific receptors have become the focus of much attention. APC-induced signaling can promote multiple cytoprotective actions which can minimize injuries in various preclinical animal injury models. Remarkably, pharmacologic therapy using APC demonstrates substantial neuroprotective effects in various murine injury models, including ischemic stroke. This review summarizes the molecules that are central to the protein C pathways, the relationship of pathway deficiencies to venous thrombosis risk, and mechanisms for the beneficial effects of APC.
Figures


References
-
- Esmon CT. Inflammation and the activated protein C anticoagulant pathway. Semin.Thromb.Hemost. 2006;32:49–60. - PubMed
-
- Weiler H, Kerschen E. Modulation of sepsis outcome with variants of activated protein C. J Thromb Haemost. 2009;7:127–131. - PubMed
-
- Jackson CJ, Xue M. Activated protein C--an anticoagulant that does more than stop clots. Int.J.Biochem.Cell Biol. 2008;40:2692–2697. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

