Species-Specific Flight Styles of Flies are Reflected in the Response Dynamics of a Homolog Motion-Sensitive Neuron
- PMID: 22485089
- PMCID: PMC3307035
- DOI: 10.3389/fnint.2012.00011
Species-Specific Flight Styles of Flies are Reflected in the Response Dynamics of a Homolog Motion-Sensitive Neuron
Abstract
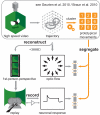
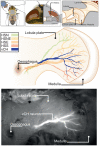
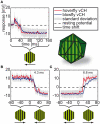
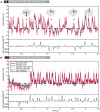
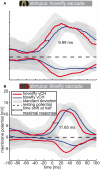
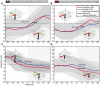
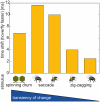
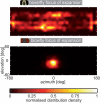
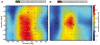
Hoverflies and blowflies have distinctly different flight styles. Yet, both species have been shown to structure their flight behavior in a way that facilitates extraction of 3D information from the image flow on the retina (optic flow). Neuronal candidates to analyze the optic flow are the tangential cells in the third optical ganglion - the lobula complex. These neurons are directionally selective and integrate the optic flow over large parts of the visual field. Homolog tangential cells in hoverflies and blowflies have a similar morphology. Because blowflies and hoverflies have similar neuronal layout but distinctly different flight behaviors, they are an ideal substrate to pinpoint potential neuronal adaptations to the different flight styles. In this article we describe the relationship between locomotion behavior and motion vision on three different levels: (1) We compare the different flight styles based on the categorization of flight behavior into prototypical movements. (2) We measure the species-specific dynamics of the optic flow under naturalistic flight conditions. We found the translational optic flow of both species to be very different. (3) We describe possible adaptations of a homolog motion-sensitive neuron. We stimulate this cell in blowflies (Calliphora) and hoverflies (Eristalis) with naturalistic optic flow generated by both species during free flight. The characterized hoverfly tangential cell responds faster to transient changes in the optic flow than its blowfly homolog. It is discussed whether and how the different dynamical response properties aid optic flow analysis.
Keywords: calliphora vicina; eristalis tenax; lobula tangential cells; motion vision; optic flow; prototypical movements; ventral centrifugal horizontal cell.
Figures


References
LinkOut - more resources
Full Text Sources

