Fstl1 antagonizes BMP signaling and regulates ureter development
- PMID: 22485132
- PMCID: PMC3317656
- DOI: 10.1371/journal.pone.0032554
Fstl1 antagonizes BMP signaling and regulates ureter development
Abstract
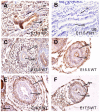
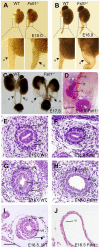
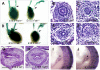
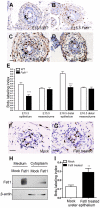
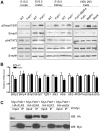
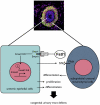
Bone morphogenetic protein (BMP) signaling pathway plays important roles in urinary tract development although the detailed regulation of its activity in this process remains unclear. Here we report that follistatin-like 1 (Fstl1), encoding a secreted extracellular glycoprotein, is expressed in developing ureter and antagonizes BMP signaling activity. Mouse embryos carrying disrupted Fstl1 gene displayed prominent hydroureter arising from proximal segment and ureterovesical junction defects. These defects were associated with significant reduction in ureteric epithelial cell proliferation at E15.5 and E16.5 as well as absence of subepithelial ureteral mesenchymal cells in the urinary tract at E16.5 and E18.5. At the molecular level, increased BMP signaling was found in Fstl1 deficient ureters, indicated by elevated pSmad1/5/8 activity. In vitro study also indicated that Fstl1 can directly bind to ALK6 which is specifically expressed in ureteric epithelial cells in developing ureter. Furthermore, Sonic hedgehog (SHH) signaling, which is crucial for differentiation of ureteral subepithelial cell proliferation, was also impaired in Fstl1(-/-) ureter. Altogether, our data suggest that Fstl1 is essential in maintaining normal ureter development by antagonizing BMP signaling.
Conflict of interest statement
Figures

Similar articles
-
Bone morphogenetic protein 4 regulates the budding site and elongation of the mouse ureter.J Clin Invest. 2000 Apr;105(7):863-73. doi: 10.1172/JCI8256. J Clin Invest. 2000. PMID: 10749566 Free PMC article.
-
Follistatin-like 1 (Fstl1) is a bone morphogenetic protein (BMP) 4 signaling antagonist in controlling mouse lung development.Proc Natl Acad Sci U S A. 2011 Apr 26;108(17):7058-63. doi: 10.1073/pnas.1007293108. Epub 2011 Apr 11. Proc Natl Acad Sci U S A. 2011. PMID: 21482757 Free PMC article.
-
Follistatin-like 1 in vertebrate development.Birth Defects Res C Embryo Today. 2013 Mar;99(1):61-9. doi: 10.1002/bdrc.21030. Birth Defects Res C Embryo Today. 2013. PMID: 23723173 Review.
-
The BMP antagonist follistatin-like 1 is required for skeletal and lung organogenesis.PLoS One. 2011;6(8):e22616. doi: 10.1371/journal.pone.0022616. Epub 2011 Aug 3. PLoS One. 2011. PMID: 21826198 Free PMC article.
-
Bone morphogenetic proteins.Growth Factors. 2004 Dec;22(4):233-41. doi: 10.1080/08977190412331279890. Growth Factors. 2004. PMID: 15621726 Review.
Cited by
-
Large genomic fragment deletions and insertions in mouse using CRISPR/Cas9.PLoS One. 2015 Mar 24;10(3):e0120396. doi: 10.1371/journal.pone.0120396. eCollection 2015. PLoS One. 2015. PMID: 25803037 Free PMC article.
-
Combinatory microarray and SuperSAGE analyses identify pairing-dependently transcribed genes in Schistosoma mansoni males, including follistatin.PLoS Negl Trop Dis. 2013 Nov 7;7(11):e2532. doi: 10.1371/journal.pntd.0002532. eCollection 2013 Nov. PLoS Negl Trop Dis. 2013. PMID: 24244773 Free PMC article.
-
Identifying pathogenic variants in the Follistatin-like 1 gene (FSTL1) in patients with skeletal and atrioventricular valve disorders.Mol Genet Genomic Med. 2019 Apr;7(4):e00567. doi: 10.1002/mgg3.567. Epub 2019 Feb 5. Mol Genet Genomic Med. 2019. PMID: 30722102 Free PMC article.
-
Smad4 regulates ureteral smooth muscle cell differentiation during mouse embryogenesis.PLoS One. 2014 Aug 15;9(8):e104503. doi: 10.1371/journal.pone.0104503. eCollection 2014. PLoS One. 2014. PMID: 25127126 Free PMC article.
-
Structural and functional study of FK domain of Fstl1.Protein Sci. 2019 Oct;28(10):1819-1829. doi: 10.1002/pro.3696. Epub 2019 Aug 9. Protein Sci. 2019. PMID: 31351024 Free PMC article.
References
-
- Pope JCt, Brock JW, Adams MC, Stephens FD, Ichikawa I. How they begin and how they end: classic and new theories for the development and deterioration of congenital anomalies of the kidney and urinary tract, CAKUT. J Am Soc Nephrol. 1999;10:2018–2028. - PubMed
-
- Miyazaki Y, Ichikawa I. Ontogeny of congenital anomalies of the kidney and urinary tract, CAKUT. Pediatr Int. 2003;45:598–604. - PubMed
-
- Vainio S, Lin Y. Coordinating early kidney development: lessons from gene targeting. Nat Rev Genet. 2002;3:533–543. - PubMed
-
- Uetani N, Bouchard M. Plumbing in the embryo: developmental defects of the urinary tracts. Clin Genet. 2009;75:307–317. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

