Haploidentical allogeneic hematopoietic cell transplantation in adults using CD3/CD19 depletion and reduced intensity conditioning: a phase II study
- PMID: 22491731
- PMCID: PMC3487553
- DOI: 10.3324/haematol.2011.059378
Haploidentical allogeneic hematopoietic cell transplantation in adults using CD3/CD19 depletion and reduced intensity conditioning: a phase II study
Abstract
Background: We report a prospective multicenter phase II study of haploidentical hematopoietic stem cell transplantation using CD3/CD19-depleted grafts after reduced intensity conditioning with fludarabine, thiotepa, melphalan and OKT-3.
Design and methods: Sixty-one adults with a median age of 46 years (range 19-65 years) have been enrolled. Diagnoses were acute myeloid leukemia (n=38), acute lymphoblastic leukemia (n=8), non-Hodgkin's lymphoma (n=6), myeloma (n=4), chronic myeloid leukemia (n=3), chronic lymphatic leukemia (n=1) and myelodysplastic syndrome (n=1). Patients were considered high risk because of refractory disease (n=18), cytogenetics (n=6), complete remission (≥ 2) (n=9), chemosensitive relapse in partial remission (n=4) or relapse after prior hematopoietic stem cell transplantation (n=15 allogeneic, n=8 autologous, n=1 both). At haploidentical hematopoietic stem cell transplantation, 30 patients were in complete remission and 31 in partial remission. Grafts contained a median of 7.0 × 10(6) (range 3.2-22) CD34(+) cells/kg, 4.2 × 10(4) (range 0.6-44) CD3(+) T cells/kg and 2.7 × 10(7) (range 0.00-37.3) CD56(+) cells/kg.
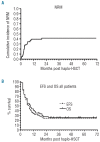
Results: Engraftment was rapid with a median of 12 days to granulocytes more than 0.5 × 10(9)/L (range 9-50 days) and 11 days to platelets more than 20 × 10(9) (range 7-38 days). Incidence of grade IIIV acute graft-versus-host-disease and chronic graft-versus-host-disease was 46% and 18%, respectively. Non-relapse mortality on Day 100 was 23% and 42% at two years. Cumulative incidence of relapse/progression at two years was 31%. Kaplan-Meier estimated 1-year and 2-year overall survival with median follow up of 869 days (range 181-1932) is 41% and 28%, respectively.
Conclusions: This regimen allows successful haploidentical hematopoietic stem cell transplantation with reduced intensity conditioning in high-risk patients lacking a suitable donor. (clinicaltrials.gov identifier:NCT00202917).
Figures


References
-
- Martin PJ. Overview of Marrow Transplantation Immunology. In: Blume KG, Forman SJ, Appelbaum FR, eds Thomas' Hematopoietic cell transplantation. Malden, MA: Blackwell Science, Inc., 2004:16-30
-
- Anasetti C, Beatty PG, Storb R, Martin PJ, Mori M, Sanders JE, et al. Effect of HLA incompatibility on graft-versus-host disease, relapse, and survival after marrow transplantation for patients with leukemia or lymphoma. Hum Immunol. 1990;29(2):79-91 - PubMed
-
- Aversa F, Tabilio A, Velardi A, Cunningham I, Terenzi A, Falzetti F, et al. Treatment of high-risk acute leukemia with T-celldepleted stem cells from related donors with one fully mismatched HLA haplotype. N Engl J Med. 1998;339(17):1186-93 - PubMed
-
- Aversa F, Tabilio A, Terenzi A, Velardi A, Falzetti F, Giannoni C, et al. Successful engraftment of T-cell-depleted haploidentical “three-loci” incompatible transplants in leukemia patients by addition of recombinant human granulocyte colony-stimulating factor-mobilized peripheral blood progenitor cells to bone marrow inoculum. Blood. 1994;84(11):3948-55 - PubMed
-
- Aversa F, Terenzi A, Tabilio A, Falzetti F, Carotti A, Ballanti S, et al. Full haplotypemismatched hematopoietic stem-cell transplantation: a phase II study in patients with acute leukemia at high risk of relapse. J Clin Oncol. 2005;23(15):3447-54 - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials

