Extracellular histones inhibit efferocytosis
- PMID: 22495510
- PMCID: PMC3409281
- DOI: 10.2119/molmed.2012.00005
Extracellular histones inhibit efferocytosis
Abstract
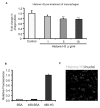
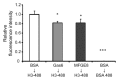
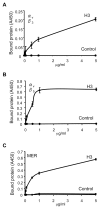
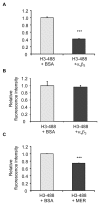
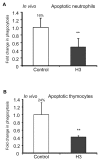
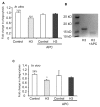
The uptake and clearance of apoptotic cells by macrophages and other phagocytic cells, a process called efferocytosis, is a major component in the resolution of inflammation. Increased concentrations of extracellular histones are found during acute inflammatory states and appear to contribute to organ system dysfunction and mortality. In these studies, we examined the potential role of histones in modulating efferocytosis. We found that phagocytosis of apoptotic neutrophils or thymocytes by macrophages was significantly diminished in the presence of histones H3 or H4, but not histone H1. Histone H3 demonstrated direct binding to macrophages, an effect that was diminished by preincubation of macrophages with the opsonins growth arrest-specific gene 6 (Gas6) and milk fat globule-epidermal growth factor (EGF) 8 (MFG-E8). Incubation of histone H3 with soluble α(v)β₅ integrin and Mer, but not with α(v)β₃, diminished its binding to macrophages. Phagocytosis of apoptotic cells by alveolar macrophages in vivo was diminished in the presence of histone H3. Incubation of histone H3 with activated protein C, a treatment that degrades histones, abrogated its inhibitory effects on efferocytosis under both in vitro and in vivo conditions. The present studies demonstrate that histones have inhibitory effects on efferocytosis, suggesting a new mechanism by which extracellular histones contribute to acute inflammatory processes and tissue injury.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

