The regulation of K- and L-cell activity by GLUT2 and the calcium-sensing receptor CasR in rat small intestine
- PMID: 22495587
- PMCID: PMC3448156
- DOI: 10.1113/jphysiol.2011.223800
The regulation of K- and L-cell activity by GLUT2 and the calcium-sensing receptor CasR in rat small intestine
Abstract
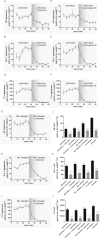
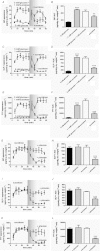
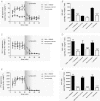
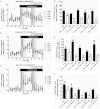
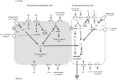
Intestinal enteroendocrine cells (IECs) secrete gut peptides in response to both nutrients and non-nutrients. Glucose and amino acids both stimulate gut peptide secretion. Our hypothesis was that the facilitative glucose transporter, GLUT2, could act as a glucose sensor and the calcium-sensing receptor, CasR, could detect amino acids in the intestine to modify gut peptide secretion. We used isolated loops of rat small intestine to study the secretion of gluco-insulinotropic peptide (GIP), glucagon-like peptide-1 (GLP-1) and peptide tyrosine tyrosine (PYY) secretion stimulated by luminal perfusion of nutrients or bile acid. Inhibition of the sodium-dependent glucose cotransporter 1 (SGLT1) with phloridzin partially inhibited GIP, GLP-1 and PYY secretion by 45%, suggesting another glucose sensor might be involved in modulating peptide secretion. The response was completely abolished in the presence of the GLUT2 inhibitors phloretin or cytochalasin B. Given that GLUT2 modified gut peptide secretion stimulated by glucose, we investigated whether it was involved in the secretion of gut peptide by other gut peptide secretagogues. Phloretin completely abolished gut peptide secretion stimulated by artificial sweetener (sucralose), dipeptide (glycylsarcosine), lipid (oleoylethanolamine), short chain fatty acid (propionate) and major rat bile acid (taurocholate) indicating a fundamental position for GLUT2 in the gut peptide secretory mechanism. We investigated how GLUT2 was able to influence gut peptide secretion mediated by a diverse range of stimulators and discovered that GLUT2 affected membrane depolarisation through the closure of K+(ATP)-sensitive channels. In the absence of SGLT1 activity (or presence of phloridzin), the secretion of GIP, GLP-1 and PYY was sensitive to K+(ATP)-sensitive channel modulators tolbutamide and diazoxide. L-amino acids phenylalanine (Phe), tryptophan (Trp), asparagine (Asn), arginine (Arg) and glutamine (Gln) also stimulated GIP, GLP-1 and PYY secretion, which was completely abolished when extracellular Ca2+ was absent. The gut peptide response stimulated by the amino acids was also blocked by the CasR inhibitor Calhex 231 and augmented by the CasR agonist NPS-R568. GLUT2 and CasR regulate K- and L-cell activity in response to nutrient and non-nutrient stimuli.
Figures




Comment in
-
Intestinal incretin responses to increased GLUT2 expression--Chacun à son goût.J Physiol. 2012 Jun 15;590(12):2825-6. doi: 10.1113/jphysiol.2012.233445. J Physiol. 2012. PMID: 22707587 Free PMC article. No abstract available.
References
-
- Adibi SA, Modesto TA, Morse EL, Amin PM. Amino acid levels in plasma, liver, and skeletal muscle during protein deprivation. Am J Physiol. 1973;225:408–414. - PubMed
-
- Auchere D, Tardivel S, Gounelle JC, Lacour B. Stimulation of ileal transport of calcium by sorbitol in in situ perfused loop in rats (in French) Gastroenterol Clin Biol. 1997;21:960–966. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

