Lysosomal dysfunction in a mouse model of Sandhoff disease leads to accumulation of ganglioside-bound amyloid-β peptide
- PMID: 22496568
- PMCID: PMC6622109
- DOI: 10.1523/JNEUROSCI.4860-11.2012
Lysosomal dysfunction in a mouse model of Sandhoff disease leads to accumulation of ganglioside-bound amyloid-β peptide
Abstract
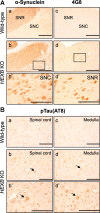
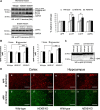
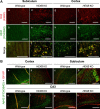
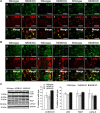
Alterations in the lipid composition of endosomal-lysosomal membranes may constitute an early event in Alzheimer's disease (AD) pathogenesis. In this study, we investigated the possibility that GM2 ganglioside accumulation in a mouse model of Sandhoff disease might be associated with the accumulation of intraneuronal and extracellular proteins commonly observed in AD. Our results show intraneuronal accumulation of amyloid-β peptide (Aβ)-like, α-synuclein-like, and phospho-tau-like immunoreactivity in the brains of β-hexosaminidase knock-out (HEXB KO) mice. Biochemical and immunohistochemical analyses confirmed that at least some of the intraneuronal Aβ-like immunoreactivity (iAβ-LIR) represents amyloid precursor protein C-terminal fragments (APP-CTFs) and/or Aβ. In addition, we observed increased levels of Aβ40 and Aβ42 peptides in the lipid-associated fraction of HEXB KO mouse brains, and intraneuronal accumulation of ganglioside-bound Aβ (GAβ) immunoreactivity in a brain region-specific manner. Furthermore, α-synuclein and APP-CTFs and/or Aβ were found to accumulate in different regions of the substantia nigra, indicating different mechanisms of accumulation or turnover pathways. Based on the localization of the accumulated iAβ-LIR to endosomes, lysosomes, and autophagosomes, we conclude that a significant accumulation of iAβ-LIR may be associated with the lysosomal-autophagic turnover of Aβ and fragments of APP-containing Aβ epitopes. Importantly, intraneuronal GAβ immunoreactivity, a proposed prefibrillar aggregate found in AD, was found to accumulate throughout the frontal cortices of postmortem human GM1 gangliosidosis, Sandhoff disease, and Tay-Sachs disease brains. Together, these results establish an association between the accumulation of gangliosides, autophagic vacuoles, and the intraneuronal accumulation of proteins associated with AD.
Figures



References
-
- Bahr BA, Bendiske J. The neuropathogenic contributions of lysosomal dysfunction. J Neurochem. 2002;83:481–489. - PubMed
-
- Barelli H, Lebeau A, Vizzavona J, Delaere P, Chevallier N, Drouot C, Marambaud P, Ancolio K, Buxbaum JD, Khorkova O, Heroux J, Sahasrabudhe S, Martinez J, Warter JM, Mohr M, Checler F. Characterization of new polyclonal antibodies specific for 40 and 42 amino acid-long amyloid beta peptides: their use to examine the cell biology of presenilins and the immunohistochemistry of sporadic Alzheimer's disease and cerebral amyloid angiopathy cases. Mol Med. 1997;3:695–707. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous
