Nano-advantage in enhanced drug delivery with biodegradable nanoparticles: contribution of reduced clearance
- PMID: 22498894
- PMCID: PMC3382836
- DOI: 10.1124/dmd.112.044925
Nano-advantage in enhanced drug delivery with biodegradable nanoparticles: contribution of reduced clearance
Abstract
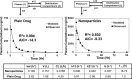
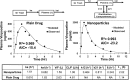
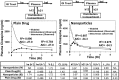
The aim of this study was to investigate the contribution of reduced apparent clearance to the enhanced exposure reported for biodegradable nanoparticles after extravascular and intravascular routes of administration. Plasma concentration profiles for drug and nanoparticle formulations after administration by intravenous, intraduodenal, and oral routes were extracted from the literature. Data were fit to pharmacokinetic models using BOOMER. The compartmental pharmacokinetic analysis of literature data for six drugs (camptothecin, 9-nitrocamptothecin, epirubicin, vinpocetine, clozapine, and cyclosporine) showed that the encapsulation of drug molecules in nanoparticles significantly reduced the apparent clearance and prolonged the apparent circulation half-life compared with those for the plain drug. Positively charged nanoparticles assessed in this study had lower apparent clearance, lower elimination rate constant values, and longer apparent circulation half-life than neutral and negatively charged nanoparticles. After oral administration, a reduction in apparent clearance contributed substantially to elevations in plasma drug exposure with nanoparticles. For the drugs and delivery systems examined, the nano-advantage in drug delivery enhancement can be explained, in part, by reduced clearance.
Figures




References
-
- Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB. (2007) Bioavailability of curcumin: problems and promises. Mol Pharm 4:807–818 - PubMed
-
- Andresen TL, Jensen SS, Jørgensen K. (2005) Advanced strategies in liposomal cancer therapy: problems and prospects of active and tumor specific drug release. Prog Lipid Res 44:68–97 - PubMed
-
- Bargoni A, Cavalli R, Caputo O, Fundarò A, Gasco MR, Zara GP. (1998) Solid lipid nanoparticles in lymph and plasma after duodenal administration to rats. Pharm Res 15:745–750 - PubMed
-
- Bawa R. (2008) Nanoparticle-based therapeutics in humans: a survey. Nanotechnol Law Bus 5:135–155
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

