Contribution of impaired mitochondrial autophagy to cardiac aging: mechanisms and therapeutic opportunities
- PMID: 22499902
- PMCID: PMC3353545
- DOI: 10.1161/CIRCRESAHA.111.246108
Contribution of impaired mitochondrial autophagy to cardiac aging: mechanisms and therapeutic opportunities
Abstract
The prevalence of cardiovascular disease increases with advancing age. Although long-term exposure to cardiovascular risk factors plays a major role in the etiopathogenesis of cardiovascular disease, intrinsic cardiac aging enhances the susceptibility to developing heart pathologies in late life. The progressive decline of cardiomyocyte mitochondrial function is considered a major mechanism underlying heart senescence. Damaged mitochondria not only produce less ATP but also generate increased amounts of reactive oxygen species and display a greater propensity to trigger apoptosis. Given the postmitotic nature of cardiomyocytes, the efficient removal of dysfunctional mitochondria is critical for the maintenance of cell homeostasis, because damaged organelles cannot be diluted by cell proliferation. The only known mechanism whereby mitochondria are turned over is through macroautophagy. The efficiency of this process declines with advancing age, which may play a critical role in heart senescence and age-related cardiovascular disease. The present review illustrates the putative mechanisms whereby alterations in the autophagic removal of damaged mitochondria intervene in the process of cardiac aging and in the pathogenesis of specific heart diseases that are especially prevalent in late life (eg, left ventricular hypertrophy, ischemic heart disease, heart failure, and diabetic cardiomyopathy). Interventions proposed to counteract cardiac aging through improvements in macroautophagy (eg, calorie restriction and calorie restriction mimetics) are also presented.
Figures

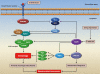
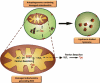
References
-
- Lloyd-Jones D, Adams R, Carnethon M, De SG, Ferguson TB, Flegal K, Ford E, Furie K, Go A, Greenlund K, Haase N, Hailpern S, Ho M, Howard V, Kissela B, Kittner S, Lackland D, Lisabeth L, Marelli A, McDermott M, Meigs J, Mozaffarian D, Nichol G, O'Donnell C, Roger V, Rosamond W, Sacco R, Sorlie P, Stafford R, Steinberger J, Thom T, Wasserthiel-Smoller S, Wong N, Wylie-Rosett J, Hong Y. Heart disease and stroke statistics--2009 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2009;119:480–6. - PubMed
-
- Odding E, Valkenburg HA, Stam HJ, Hofman A. Determinants of locomotor disability in people aged 55 years and over: the Rotterdam Study. Eur J Epidemiol. 2001;17:1033–41. - PubMed
-
- Newman AB, Arnold AM, Naydeck BL, Fried LP, Burke GL, Enright P, Gottdiener J, Hirsch C, O'Leary D, Tracy R. “Successful aging”: effect of subclinical cardiovascular disease. Arch Intern Med. 2003;163:2315–22. - PubMed
-
- Lakatta EG. Heart aging: a fly in the ointment? Circ Res. 2001;88:984–6. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

