All-trans-retinoic acid ameliorated high fat diet-induced atherosclerosis in rabbits by inhibiting platelet activation and inflammation
- PMID: 22505807
- PMCID: PMC3303861
- DOI: 10.1155/2012/259693
All-trans-retinoic acid ameliorated high fat diet-induced atherosclerosis in rabbits by inhibiting platelet activation and inflammation
Abstract
Background: All-trans-retinoic acid (atRA) is effective for many proliferative diseases. We investigated the protective effects of atRA against atherosclerosis.
Methods: Rabbits were randomly allocated to receive basal diet or an HFD for 4 weeks. HFD group then received rosuvastatin (3 mg/day), atRA (5 mg/kg/day), or the same volume of vehicle, respectively, for next 8 weeks.
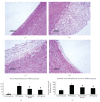
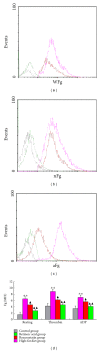
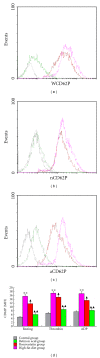
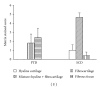
Results: HFD group showed increases in plasma lipids and aortic plaque formation. P-selectin expression and fibrinogen binding on platelets or deposition on the intima of the aorta also increased significantly as did the levels of TNF-α, IL-6, and fibrinogen in plasma. After 8 weeks of treatment with atRA, there was a significant decrease in plasma lipids and improvement in aortic lesions. AtRA also inhibited the expression of P-selectin and fibrinogen binding on platelets and deposition on the intima of the aorta.
Conclusion: AtRA can ameliorate HFD-induced AS in rabbits by inhibiting platelet activation and inflammation.
Figures

Similar articles
-
All-trans retinoic acid effectively reduces atheroma plaque size in a rabbit model of high-fat-induced atherosclerosis.Adv Clin Exp Med. 2018 Dec;27(12):1631-1636. doi: 10.17219/acem/74552. Adv Clin Exp Med. 2018. PMID: 30048051
-
ATRA improves endothelial dysfunction in atherosclerotic rabbits by decreasing CAV‑1 expression and enhancing eNOS activity.Mol Med Rep. 2018 May;17(5):6796-6802. doi: 10.3892/mmr.2018.8647. Epub 2018 Feb 27. Mol Med Rep. 2018. PMID: 29488619
-
All-trans-retinoic acid ameliorates atherosclerosis, promotes perivascular adipose tissue browning, and increases adiponectin production in Apo-E mice.Sci Rep. 2021 Feb 24;11(1):4451. doi: 10.1038/s41598-021-83939-x. Sci Rep. 2021. PMID: 33627760 Free PMC article.
-
Fibrinogen and P-selectin expression in atherosclerosis model of Sprague Dawley rat.Chin Med J (Engl). 2011 Nov;124(22):3768-72. Chin Med J (Engl). 2011. PMID: 22340239
-
All-trans retinoic acid reduces intimal thickening after balloon angioplasty in atherosclerotic rabbits.Chin Med J (Engl). 1999 Feb;112(2):121-3. Chin Med J (Engl). 1999. PMID: 11593575
Cited by
-
Inhibition of thrombin, an unexplored function of retinoic acid.Biochem Biophys Rep. 2019 Apr 25;18:100636. doi: 10.1016/j.bbrep.2019.100636. eCollection 2019 Jul. Biochem Biophys Rep. 2019. PMID: 31049420 Free PMC article.
-
Myosin light chain kinase inhibitor ML7 improves vascular endothelial dysfunction via tight junction regulation in a rabbit model of atherosclerosis.Mol Med Rep. 2015 Sep;12(3):4109-4116. doi: 10.3892/mmr.2015.3973. Epub 2015 Jun 22. Mol Med Rep. 2015. PMID: 26096176 Free PMC article.
-
Chronic inflammation and vascular cell plasticity in atherosclerosis.Nat Cardiovasc Res. 2024 Dec;3(12):1408-1423. doi: 10.1038/s44161-024-00569-y. Epub 2024 Dec 9. Nat Cardiovasc Res. 2024. PMID: 39653823 Review.
-
Sunflower oil supplementation has proinflammatory effects and does not reverse insulin resistance in obesity induced by high-fat diet in C57BL/6 mice.J Biomed Biotechnol. 2012;2012:945131. doi: 10.1155/2012/945131. Epub 2012 Aug 26. J Biomed Biotechnol. 2012. PMID: 22988427 Free PMC article.
-
Single-Cell Genomics Reveals a Novel Cell State During Smooth Muscle Cell Phenotypic Switching and Potential Therapeutic Targets for Atherosclerosis in Mouse and Human.Circulation. 2020 Nov 24;142(21):2060-2075. doi: 10.1161/CIRCULATIONAHA.120.048378. Epub 2020 Sep 23. Circulation. 2020. PMID: 32962412 Free PMC article.
References
-
- Axel DI, Frigge A, Dittmann J, et al. All-trans retinoic acid regulates proliferation, migration, differentiation, and extracellular matrix turnover of human arterial smooth muscle cells. Cardiovascular Research. 2001;49(4):851–862. - PubMed
-
- Miano JM, Topouzis S, Majesky MW, Olson EN. Retinoid receptor expression and all-trans retinoic acid-mediated growth inhibition in vascular smooth muscle cells. Circulation. 1996;93(10):1886–1895. - PubMed
-
- Ou H, Haendeler J, Aebly MR, et al. Retinoic acid-induced tissue transglutaminase and apoptosis in vascular smooth muscle cells. Circulation Research. 2000;87(10):881–887. - PubMed
-
- Wiegman PJ, Barry WL, McPherson JA, et al. All-trans-retinoic acid limits restenosis after balloon angioplasty in the focally atherosclerotic rabbit: a favorable effect on vessel remodeling. Arteriosclerosis, Thrombosis, and Vascular Biology. 2000;20(1):89–95. - PubMed
-
- Herdeg C, Oberhoff M, Baumbach A, et al. Effects of local all-trans-retinoic acid delivery on experimental atherosclerosis in the rabbit carotid artery. Cardiovascular Research. 2003;57(2):544–553. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

