Virus-drug interactions--molecular insight into immunosuppression and HCV
- PMID: 22508161
- PMCID: PMC7097508
- DOI: 10.1038/nrgastro.2012.67
Virus-drug interactions--molecular insight into immunosuppression and HCV
Abstract
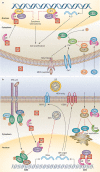
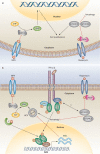
Liver transplantation is an effective treatment for end-stage liver disease that is attributable to chronic HCV infection. However, long-term outcomes are compromised by universal virological recurrence in the graft. Reinfection that occurs after transplantation has increased resistance to current interferon-based antiviral therapy and often leads to accelerated development of cirrhosis. Important risk factors for severe HCV recurrence are linked to immunosuppression. Owing to the lack of good randomized, controlled trials, the optimal choice of immunosuppressants is still debated. By contrast, much progress has been made in the understanding of HCV biology and the antiviral action of interferons. These new insights have greatly expanded our knowledge of the molecular interplay between HCV and immunosuppressive drugs. In this article, we explore the effect of different immunosuppressants on the complex cellular events involved in HCV infection and interferon signalling. Potential implications for clinical practice and future drug development are discussed.
Conflict of interest statement
H. J. Metselaar has been a consultant and speaker for Astrellas and Novarits. He has also received grant/research support from Astrellas. The other authors declare no competing interests.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

