The extracellular signal-regulated kinase 3 (mitogen-activated protein kinase 6 [MAPK6])-MAPK-activated protein kinase 5 signaling complex regulates septin function and dendrite morphology
- PMID: 22508986
- PMCID: PMC3434500
- DOI: 10.1128/MCB.06633-11
The extracellular signal-regulated kinase 3 (mitogen-activated protein kinase 6 [MAPK6])-MAPK-activated protein kinase 5 signaling complex regulates septin function and dendrite morphology
Abstract
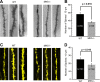
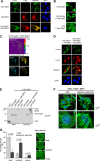
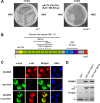
Mitogen-activated protein kinase-activated protein (MAPKAP) kinase 5 (MK5) deficiency is associated with reduced extracellular signal-regulated kinase 3 (ERK3) (mitogen-activated protein kinase 6) levels, hence we utilized the MK5 knockout mouse model to analyze the physiological functions of the ERK3/MK5 signaling module. MK5-deficient mice displayed impaired dendritic spine formation in mouse hippocampal neurons in vivo. We performed large-scale interaction screens to understand the neuronal functions of the ERK3/MK5 pathway and identified septin7 (Sept7) as a novel interacting partner of ERK3. ERK3/MK5/Sept7 form a ternary complex, which can phosphorylate the Sept7 regulators Binders of Rho GTPases (Borgs). In addition, the brain-specific nucleotide exchange factor kalirin-7 (Kal7) was identified as an MK5 interaction partner and substrate protein. In transfected primary neurons, Sept7-dependent dendrite development and spine formation are stimulated by the ERK3/MK5 module. Thus, the regulation of neuronal morphogenesis is proposed as the first physiological function of the ERK3/MK5 signaling module.
Figures




Similar articles
-
Activation of MK5/PRAK by the atypical MAP kinase ERK3 defines a novel signal transduction pathway.EMBO J. 2004 Dec 8;23(24):4780-91. doi: 10.1038/sj.emboj.7600489. Epub 2004 Dec 2. EMBO J. 2004. PMID: 15577943 Free PMC article.
-
Docking of PRAK/MK5 to the atypical MAPKs ERK3 and ERK4 defines a novel MAPK interaction motif.J Biol Chem. 2009 Jul 17;284(29):19392-401. doi: 10.1074/jbc.M109.023283. Epub 2009 May 27. J Biol Chem. 2009. PMID: 19473979 Free PMC article.
-
Scaffolding by ERK3 regulates MK5 in development.EMBO J. 2004 Dec 8;23(24):4770-9. doi: 10.1038/sj.emboj.7600467. Epub 2004 Nov 11. EMBO J. 2004. PMID: 15538386 Free PMC article.
-
MK5: A novel regulator of cardiac fibroblast function?IUBMB Life. 2017 Oct;69(10):785-794. doi: 10.1002/iub.1677. Epub 2017 Sep 11. IUBMB Life. 2017. PMID: 28941148 Review.
-
New insights into the activation, interaction partners and possible functions of MK5/PRAK.Front Biosci (Landmark Ed). 2016 Jan 1;21(2):374-84. doi: 10.2741/4394. Front Biosci (Landmark Ed). 2016. PMID: 26709779 Review.
Cited by
-
Deubiquitinating Enzyme USP20 Regulates Extracellular Signal-Regulated Kinase 3 Stability and Biological Activity.Mol Cell Biol. 2017 Apr 14;37(9):e00432-16. doi: 10.1128/MCB.00432-16. Print 2017 May 1. Mol Cell Biol. 2017. PMID: 28167606 Free PMC article.
-
Brain Region and Isoform-Specific Phosphorylation Alters Kalirin SH2 Domain Interaction Sites and Calpain Sensitivity.ACS Chem Neurosci. 2017 Jul 19;8(7):1554-1569. doi: 10.1021/acschemneuro.7b00076. Epub 2017 Apr 25. ACS Chem Neurosci. 2017. PMID: 28418645 Free PMC article.
-
Septin Assembly and Remodeling at the Cell Division Site During the Cell Cycle.Front Cell Dev Biol. 2021 Nov 25;9:793920. doi: 10.3389/fcell.2021.793920. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34901034 Free PMC article. Review.
-
Positional plasticity in regenerating Amybstoma mexicanum limbs is associated with cell proliferation and pathways of cellular differentiation.BMC Dev Biol. 2015 Nov 23;15:45. doi: 10.1186/s12861-015-0095-4. BMC Dev Biol. 2015. PMID: 26597593 Free PMC article.
-
Kalirin as a Novel Treatment Target for Cognitive Dysfunction in Schizophrenia.CNS Drugs. 2022 Jan;36(1):1-16. doi: 10.1007/s40263-021-00884-z. Epub 2021 Dec 20. CNS Drugs. 2022. PMID: 34928485 Review.
References
-
- Aberg E, et al. 2006. Regulation of MAPK-activated protein kinase 5 activity and subcellular localization by the atypical MAPK ERK4/MAPK4. J. Biol. Chem. 281: 35499–35510 - PubMed
-
- Barral Y. 2010. Cell biology. Septins at the nexus. Science 329: 1289–1290 - PubMed
-
- Barral Y, Mansuy IM. 2007. Septins: cellular and functional barriers of neuronal activity. Curr. Biol. 17: R961–R963 - PubMed
-
- Broder YC, Katz S, Aronheim A. 1998. The ras recruitment system, a novel approach to the study of protein-protein interactions. Curr. Biol. 8: 1121–1124 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
