Effect on adherence to nicotine replacement therapy of informing smokers their dose is determined by their genotype: a randomised controlled trial
- PMID: 22509402
- PMCID: PMC3324463
- DOI: 10.1371/journal.pone.0035249
Effect on adherence to nicotine replacement therapy of informing smokers their dose is determined by their genotype: a randomised controlled trial
Abstract
Background: The behavioural impact of pharmacogenomics is untested. We tested two hypotheses concerning the behavioural impact of informing smokers their oral dose of NRT is tailored to analysis of DNA.
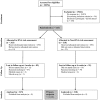
Methods and findings: We conducted an RCT with smokers in smoking cessation clinics (N = 633). In combination with NRT patch, participants were informed that their doses of oral NRT were based either on their mu-opioid receptor (OPRM1) genotype, or their nicotine dependence questionnaire score (phenotype). The proportion of prescribed NRT consumed in the first 28 days following quitting was not significantly different between groups: (68.5% of prescribed NRT consumed in genotype vs 63.6%, phenotype group, difference = 5.0%, 95% CI -0.9,10.8, p = 0.098). Motivation to make another quit attempt among those (n = 331) not abstinent at six months was not significantly different between groups (p = 0.23). Abstinence at 28 days was not different between groups (p = 0.67); at six months was greater in genotype than phenotype group (13.7% vs 7.9%, difference = 5.8%, 95% CI 1.0,10.7, p = 0.018).
Conclusions: Informing smokers their oral dose of NRT was tailored to genotype not phenotype had a small, statistically non-significant effect on 28-day adherence to NRT. Among those still smoking at six months, there was no evidence that saying NRT was tailored to genotype adversely affected motivation to make another quit attempt. Higher abstinence rate at six months in the genotype arm requires investigation.
Trial registration: Controlled-Trials.com ISRCTN14352545.
Conflict of interest statement
Figures
References
-
- Li MD. The genetics of nicotine dependence. Curr Psychiatry Rep. 2006;8:158–164. - PubMed
-
- Li MD, Cheng R, Ma JZ, Swan GE. A meta-analysis of estimated genetic and environmental effects on smoking behavior in male and female adult twins. Addiction. 2003;98:23–31. - PubMed
-
- Schnoll RA, Johnson TA, Lerman C. Genetics and smoking behavior. Curr Psychiatry Rep. 2007;9:349–357. - PubMed
-
- Munafò MR, Lerman C, Niaura R, Shields AE, Swan GE. Smoking cessation treatment: Pharmacogenetic assessment. Current Opinion in Molecular Therapeutics. 2005;7:202–208. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials