Establishment and validation of computational model for MT1-MMP dependent ECM degradation and intervention strategies
- PMID: 22511862
- PMCID: PMC3325185
- DOI: 10.1371/journal.pcbi.1002479
Establishment and validation of computational model for MT1-MMP dependent ECM degradation and intervention strategies
Abstract
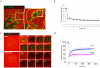
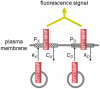
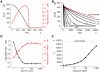
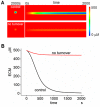
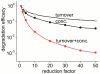
MT1-MMP is a potent invasion-promoting membrane protease employed by aggressive cancer cells. MT1-MMP localizes preferentially at membrane protrusions called invadopodia where it plays a central role in degradation of the surrounding extracellular matrix (ECM). Previous reports suggested a role for a continuous supply of MT1-MMP in ECM degradation. However, the turnover rate of MT1-MMP and the extent to which the turnover contributes to the ECM degradation at invadopodia have not been clarified. To approach this problem, we first performed FRAP (Fluorescence Recovery after Photobleaching) experiments with fluorescence-tagged MT1-MMP focusing on a single invadopodium and found very rapid recovery in FRAP signals, approximated by double-exponential plots with time constants of 26 s and 259 s. The recovery depended primarily on vesicle transport, but negligibly on lateral diffusion. Next we constructed a computational model employing the observed kinetics of the FRAP experiments. The simulations successfully reproduced our FRAP experiments. Next we inhibited the vesicle transport both experimentally, and in simulation. Addition of drugs inhibiting vesicle transport blocked ECM degradation experimentally, and the simulation showed no appreciable ECM degradation under conditions inhibiting vesicle transport. In addition, the degree of the reduction in ECM degradation depended on the degree of the reduction in the MT1-MMP turnover. Thus, our experiments and simulations have established the role of the rapid turnover of MT1-MMP in ECM degradation at invadopodia. Furthermore, our simulations suggested synergetic contributions of proteolytic activity and the MT1-MMP turnover to ECM degradation because there was a nonlinear and marked reduction in ECM degradation if both factors were reduced simultaneously. Thus our computational model provides a new in silico tool to design and evaluate intervention strategies in cancer cell invasion.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- Maquoi E, Assent D, Detilleux J, Pequeux C, Foidart JM, et al. MT1-MMP protects breast carcinoma cells against type I collagen-induced apoptosis. Oncogene. 2011;31:480–493. - PubMed
-
- Nonaka T, Nishibashi K, Itoh Y, Yana I, Seiki M. Competitive disruption of the tumor-promoting function of membrane type 1 matrix metalloproteinase/matrix metalloproteinase-14 in vivo. Mol Cancer Ther. 2005;4:1157–1166. - PubMed
-
- Taniwaki K, Fukamachi H, Komori K, Ohtake Y, Nonaka T, et al. Stroma-derived matrix metalloproteinase (MMP)-2 promotes membrane type 1-MMP-dependent tumor growth in mice. Cancer Res. 2007;67:4311–4319. - PubMed
-
- Wolf K, Friedl P. Mapping proteolytic cancer cell-extracellular matrix interfaces. Clin Exp Metastasis. 2009;26:289–298. - PubMed
-
- Yamaguchi H, Pixley F, Condeelis J. Invadopodia and podosomes in tumor invasion. Eur J Cell Biol. 2006;85:213–218. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

