LINGO-1, a transmembrane signaling protein, inhibits oligodendrocyte differentiation and myelination through intercellular self-interactions
- PMID: 22514275
- PMCID: PMC3381180
- DOI: 10.1074/jbc.M112.366179
LINGO-1, a transmembrane signaling protein, inhibits oligodendrocyte differentiation and myelination through intercellular self-interactions
Abstract
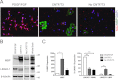
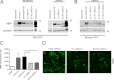
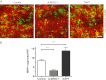
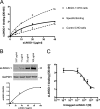
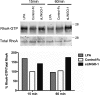
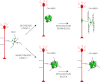
Overcoming remyelination failure is a major goal of new therapies for demyelinating diseases like multiple sclerosis. LINGO-1, a key negative regulator of myelination, is a transmembrane signaling protein expressed in both neurons and oligodendrocytes. In neurons, LINGO-1 is an integral component of the Nogo receptor complex, which inhibits axonal growth via RhoA. Because the only ligand-binding subunit of this complex, the Nogo receptor, is absent in oligodendrocytes, the extracellular signals that inhibit myelination through a LINGO-1-mediated mechanism are unknown. Here we show that LINGO-1 inhibits oligodendrocyte terminal differentiation through intercellular interactions and is capable of a self-association in trans. Consistent with previous reports, overexpression of full-length LINGO-1 inhibited differentiation of oligodendrocyte precursor cells (OPCs). Unexpectedly, treatment with a soluble recombinant LINGO-1 ectodomain also had an inhibitory effect on OPCs and decreased myelinated axonal segments in cocultures with neurons from dorsal root ganglia. We demonstrated LINGO-1-mediated inhibition of OPCs through intercellular signaling by using a surface-bound LINGO-1 construct expressed ectopically in astrocytes. Further investigation showed that the soluble LINGO-1 ectodomain can interact with itself in trans by binding to CHO cells expressing full-length LINGO-1. Finally, we observed that soluble LINGO-1 could activate RhoA in OPCs. We propose that LINGO-1 acts as both a ligand and a receptor and that the mechanism by which it negatively regulates OPC differentiation and myelination is mediated by a homophilic intercellular interaction. Disruption of this protein-protein interaction could lead to a decrease of LINGO-1 inhibition and an increase in myelination.
Figures


References
-
- Mi S., Miller R. H., Lee X., Scott M. L., Shulag-Morskaya S., Shao Z., Chang J., Thill G., Levesque M., Zhang M., Hession C., Sah D., Trapp B., He Z., Jung V., McCoy J. M., Pepinsky R. B. (2005) LINGO-1 negatively regulates myelination by oligodendrocytes. Nat. Neurosci. 8, 745–751 - PubMed
-
- Brinkmann B. G., Agarwal A., Sereda M. W., Garratt A. N., Müller T., Wende H., Stassart R. M., Nawaz S., Humml C., Velanac V., Radyushkin K., Goebbels S., Fischer T. M., Franklin R. J., Lai C., Ehrenreich H., Birchmeier C., Schwab M. H., Nave K. A. (2008) Neuregulin-1/ErbB signaling serves distinct functions in myelination of the peripheral and central nervous system. Neuron 59, 581–595 - PMC - PubMed
-
- Taveggia C., Thaker P., Petrylak A., Caporaso G. L., Toews A., Falls D. L., Einheber S., Salzer J. L. (2008) Type III neuregulin-1 promotes oligodendrocyte myelination. Glia 56, 284–293 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

