How I treat acquired aplastic anemia
- PMID: 22517900
- PMCID: PMC3418715
- DOI: 10.1182/blood-2011-12-274019
How I treat acquired aplastic anemia
Abstract
Survival in severe aplastic anemia (SAA) has markedly improved in the past 4 decades because of advances in hematopoietic stem cell transplantation, immunosuppressive biologics and drugs, and supportive care. However, management of SAA patients remains challenging, both acutely in addressing the immediate consequences of pancytopenia and in the long term because of the disease's natural history and the consequences of therapy. Recent insights into pathophysiology have practical implications. We review key aspects of differential diagnosis, considerations in the choice of first- and second-line therapies, and the management of patients after immunosuppression, based on both a critical review of the recent literature and our large personal and research protocol experience of bone marrow failure in the Hematology Branch of the National Heart, Lung, and Blood Institute.
Figures
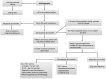
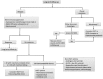
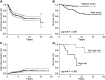
References
-
- Marsh JC, Ball SE, Cavenagh J, et al. Guidelines for the diagnosis and management of aplastic anaemia. Br J Haematol. 2009;147(1):43–70. - PubMed
-
- Young NS, Maciejewski JP. Aplastic Anemia in Hoffman Basic Principles and Practice. 5th Ed. Philadelphia, PA: Churchill Livingston Elsevier; 2009.
-
- Bloom ML, Wolk AG, Simon-Stoos KL, Bard JS, Chen J, Young NS. A mouse model of lymphocyte infusion-induced bone marrow failure. Exp Hematol. 2004;32(12):1163–1172. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

