Prenatal stress causes alterations in the morphology of microglia and the inflammatory response of the hippocampus of adult female mice
- PMID: 22520439
- PMCID: PMC3409032
- DOI: 10.1186/1742-2094-9-71
Prenatal stress causes alterations in the morphology of microglia and the inflammatory response of the hippocampus of adult female mice
Abstract
Background: Stress during fetal life increases the risk of affective and immune disorders later in life. The altered peripheral immune response caused by prenatal stress may impact on brain function by the modification of local inflammation. In this study we have explored whether prenatal stress results in alterations in the immune response in the hippocampus of female mice during adult life.
Methods: Pregnant C57BL/6 mice were subjected three times/day during 45 minutes to restraint stress from gestational Day 12 to delivery. Control non-stressed pregnant mice remained undisturbed. At four months of age, non-stressed and prenatally stressed females were ovariectomized. Fifteen days after surgery, mice received an i.p. injection of vehicle or of 5 mg/kg of lipopolysaccharide (LPS). Mice were sacrificed 20 hours later by decapitation and the brains were removed. Levels of interleukin-1β (IL1β), interleukin-6 (IL-6), tumor necrosis factor α (TNF-α), interferon γ-inducible protein 10 (IP10), and toll-like receptor 4 mRNA were assessed in the hippocampus by quantitative real-time polymerase chain reaction. Iba1 immunoreactivity was assessed by immunocytochemistry. Statistical significance was determined by one-way or two-way analysis of variance.
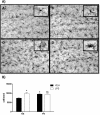
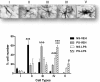
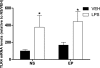
Results: Prenatal stress, per se, increased IL1β mRNA levels in the hippocampus, increased the total number of Iba1-immunoreactive microglial cells and increased the proportion of microglial cells with large somas and retracted cellular processes. In addition, prenatally stressed and non-stressed animals showed different responses to peripheral inflammation induced by systemic administration of LPS. LPS induced a significant increase in mRNA levels of IL-6, TNF-α and IP10 in the hippocampus of prenatally stressed mice but not of non-stressed animals. In addition, after LPS treatment, prenatally stressed animals showed a higher proportion of Iba1-immunoreactive cells in the hippocampus with morphological characteristics of activated microglia compared to non-stressed animals. In contrast, LPS induced similar increases in expression of IL1β and toll-like receptor 4 in both prenatally stressed and non-stressed animals.
Conclusion: These findings indicate that prenatal stress induces long-lasting modifications in the inflammatory status of the hippocampus of female mice under basal conditions and alters the immune response of the hippocampus to peripheral inflammation.
Figures

Similar articles
-
Prenatal stress increases the expression of proinflammatory cytokines and exacerbates the inflammatory response to LPS in the hippocampal formation of adult male mice.Brain Behav Immun. 2013 Feb;28:196-206. doi: 10.1016/j.bbi.2012.11.013. Epub 2012 Dec 1. Brain Behav Immun. 2013. PMID: 23207108
-
Early Life Stress Activates Glial Cells in the Hippocampus but Attenuates Cytokine Secretion in Response to an Immune Challenge in Rat Pups.Neuroimmunomodulation. 2017;24(4-5):242-255. doi: 10.1159/000485383. Epub 2018 Jan 13. Neuroimmunomodulation. 2017. PMID: 29332092
-
Prenatal stress reduces the effectiveness of the neurosteroid 3 alpha,5 alpha-THP to block kainic-acid-induced seizures.Dev Psychobiol. 1999 Apr;34(3):227-34. Dev Psychobiol. 1999. PMID: 10204098
-
Inflammation and the neural diathesis-stress hypothesis of schizophrenia: a reconceptualization.Transl Psychiatry. 2017 Feb 7;7(2):e1024. doi: 10.1038/tp.2016.278. Transl Psychiatry. 2017. PMID: 28170004 Free PMC article. Review.
-
The Impact of Systemic Inflammation on Neurodevelopment.Trends Mol Med. 2018 Sep;24(9):794-804. doi: 10.1016/j.molmed.2018.06.008. Epub 2018 Jul 11. Trends Mol Med. 2018. PMID: 30006148 Free PMC article. Review.
Cited by
-
Impact of 5-Lipoxygenase Deficiency on Dopamine-Mediated Behavioral Responses.Neurotox Res. 2024 Oct 4;42(5):42. doi: 10.1007/s12640-024-00720-4. Neurotox Res. 2024. PMID: 39365372
-
Microglial Dysregulation and Suicidality: A Stress-Diathesis Perspective.Front Psychiatry. 2020 Aug 11;11:781. doi: 10.3389/fpsyt.2020.00781. eCollection 2020. Front Psychiatry. 2020. PMID: 32848946 Free PMC article. Review.
-
Enduring neuroimmunological consequences of developmental experiences: From vulnerability to resilience.Mol Cell Neurosci. 2020 Dec;109:103567. doi: 10.1016/j.mcn.2020.103567. Epub 2020 Oct 14. Mol Cell Neurosci. 2020. PMID: 33068720 Free PMC article. Review.
-
Sex dependent impact of gestational stress on predisposition to eating disorders and metabolic disease.Mol Metab. 2018 Nov;17:1-16. doi: 10.1016/j.molmet.2018.08.005. Epub 2018 Aug 20. Mol Metab. 2018. PMID: 30174229 Free PMC article.
-
The Role of Microglia in the (Mal)adaptive Response to Traumatic Experience in an Animal Model of PTSD.Int J Mol Sci. 2022 Jun 28;23(13):7185. doi: 10.3390/ijms23137185. Int J Mol Sci. 2022. PMID: 35806185 Free PMC article.
References
-
- Barker DJP. The developmental origins of adult disease. J Am Coll Nutr. 2004;23:588S–595S. - PubMed
-
- Huizink AC, Mulder E, Buitelaar JK. Prenatal stress and risk for psychopathology: specific effects or induction of general susceptibility? Psychol Bull. 2004;130:115–142. - PubMed
-
- Maccari S, Darnaudery M, Morley-Fletcher S, Zuena AR, Cinque C, Van Reeth O. Prenatal stress and long-term consequences: implications of glucocorticoid hormones. Neurosci Biobehav Rev. 2001;27:119–127. - PubMed
-
- Maccari S, Morley-Fletcher S. Effects of prenatal restraint stress on the hypothalamus-pituitary-adrenal axis and related behavioural and neurobiological alterations. Psychoneuroendocrinology. 2007;32:S10–S15. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

