Renal disease progression in autosomal dominant polycystic kidney disease
- PMID: 22526483
- PMCID: PMC3416980
- DOI: 10.1007/s10157-012-0611-9
Renal disease progression in autosomal dominant polycystic kidney disease
Abstract
Background: Autosomal dominant polycystic kidney disease is a lifelong progressive disorder. However, how age, blood pressure, and stage of chronic kidney disease (CKD) affect the rate of kidney function deterioration is not clearly understood.
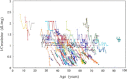
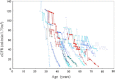
Methods: In this long-term observational case study up to 13.9 years (median observation period for slope was 3.3 years), serum creatinine was serially measured in 255 mostly adult patients. The glomerular filtration rate was estimated (eGFR) using a modified Modification of Diet in Renal Disease Study method. The total kidney volume (TKV) has been measured in 86 patients at one center since 2006.
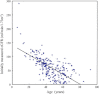
Results: As age increased, eGFR declined significantly (P < 0.0001), but the annual rate of decline of eGFR did not correlate with age or initially measured eGFR. In patients with CKD stage 1, eGFR declined at a rate which was not significantly different from other advanced CKD stages. Hypertensive patients had lower eGFR and larger TKV than normotensive patients at a young adult age. The slopes of regression lines of eGFR and TKV in relation to age were not different between high and normal blood pressure groups.
Conclusion: The declining rate of eGFR was relatively constant and did not correlate with age or eGFR after adolescence. eGFR was already low in young adult patients with hypertension. As age increased after adolescence, eGFR declined and TKV increased similarly between normal and high blood pressure groups. eGFR starts to decline in patients with normal eGFR, suggesting that the decline starts earlier than previously thought.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

