γ-Glutamyltranspeptidases: sequence, structure, biochemical properties, and biotechnological applications
- PMID: 22527720
- PMCID: PMC11115026
- DOI: 10.1007/s00018-012-0988-3
γ-Glutamyltranspeptidases: sequence, structure, biochemical properties, and biotechnological applications
Abstract
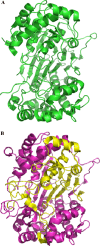
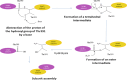
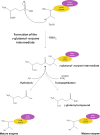
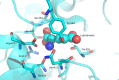
γ-Glutamyltranspeptidases (γ-GTs) are ubiquitous enzymes that catalyze the hydrolysis of γ-glutamyl bonds in glutathione and glutamine and the transfer of the released γ-glutamyl group to amino acids or short peptides. These enzymes are involved in glutathione metabolism and play critical roles in antioxidant defense, detoxification, and inflammation processes. Moreover, γ-GTs have been recently found to be involved in many physiological disorders, such as Parkinson's disease and diabetes. In this review, the main biochemical and structural properties of γ-GTs isolated from different sources, as well as their conformational stability and mechanism of catalysis, are described and examined with the aim of contributing to the discussion on their structure-function relationships. Possible applications of γ-glutamyltranspeptidases in different fields of biotechnology and medicine are also discussed.
Figures



Similar articles
-
Biochemical and structural properties of gamma-glutamyl transpeptidase from Geobacillus thermodenitrificans: an enzyme specialized in hydrolase activity.Biochimie. 2010 May;92(5):464-74. doi: 10.1016/j.biochi.2010.01.021. Epub 2010 Feb 4. Biochimie. 2010. PMID: 20138205
-
Gene cloning and protein expression of γ-glutamyltranspeptidases from Thermus thermophilus and Deinococcus radiodurans: comparison of molecular and structural properties with mesophilic counterparts.Extremophiles. 2011 Mar;15(2):259-70. doi: 10.1007/s00792-011-0355-6. Epub 2011 Feb 5. Extremophiles. 2011. PMID: 21298394
-
Exploring the unfolding mechanism of γ-glutamyltranspeptidases: the case of the thermophilic enzyme from Geobacillus thermodenitrificans.Biochim Biophys Acta. 2012 Apr;1824(4):571-7. doi: 10.1016/j.bbapap.2012.01.014. Epub 2012 Jan 31. Biochim Biophys Acta. 2012. PMID: 22322192
-
gamma-Glutamyl transpeptidase: catalytic mechanism and gene expression.Adv Enzymol Relat Areas Mol Biol. 1998;72:239-78. doi: 10.1002/9780470123188.ch7. Adv Enzymol Relat Areas Mol Biol. 1998. PMID: 9559055 Review.
-
Gamma-glutamyl compounds and their enzymatic production using bacterial gamma-glutamyltranspeptidase.Amino Acids. 2007;32(3):333-40. doi: 10.1007/s00726-006-0416-9. Epub 2006 Oct 13. Amino Acids. 2007. PMID: 17031476 Review.
Cited by
-
Bacterial Gamma-Glutamyl Transpeptidase, an Emerging Biocatalyst: Insights Into Structure-Function Relationship and Its Biotechnological Applications.Front Microbiol. 2021 Apr 9;12:641251. doi: 10.3389/fmicb.2021.641251. eCollection 2021. Front Microbiol. 2021. PMID: 33897647 Free PMC article. Review.
-
Diverse roles of low-molecular weight thiol GSH in Francisella's virulence, location sensing and GSH-stealing from host.Curr Res Microb Sci. 2023 Dec 20;6:100218. doi: 10.1016/j.crmicr.2023.100218. eCollection 2024. Curr Res Microb Sci. 2023. PMID: 38303966 Free PMC article.
-
Mutational Analysis of a Highly Conserved PLSSMXP Sequence in the Small Subunit of Bacillus licheniformis γ-Glutamyltranspeptidase.Biomolecules. 2019 Sep 19;9(9):508. doi: 10.3390/biom9090508. Biomolecules. 2019. PMID: 31546955 Free PMC article.
-
Impact of the superoxide dismutase 2 Val16Ala polymorphism on the relationship between valproic acid exposure and elevation of γ-glutamyltransferase in patients with epilepsy: a population pharmacokinetic-pharmacodynamic analysis.PLoS One. 2014 Nov 5;9(11):e111066. doi: 10.1371/journal.pone.0111066. eCollection 2014. PLoS One. 2014. PMID: 25372290 Free PMC article.
-
Gamma-glutamyl transferase and cardiovascular disease.Ann Transl Med. 2016 Dec;4(24):481. doi: 10.21037/atm.2016.12.27. Ann Transl Med. 2016. PMID: 28149843 Free PMC article. Review.
References
-
- Benlloch M, Ortega A, Ferrer P, Segarra R, Obrador E, Asensi M, Carretero J, Estrela JM. Acceleration of glutathione efflux and inhibition of gamma-glutamyltranspeptidase sensitize metastatic B16 melanoma cells to endothelium-induced cytotoxicity. J Biol Chem. 2005;280(8):6950–6959. doi: 10.1074/jbc.M408531200. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

