Protein-thiol oxidation and cell death: regulatory role of glutaredoxins
- PMID: 22530666
- PMCID: PMC3474186
- DOI: 10.1089/ars.2012.4644
Protein-thiol oxidation and cell death: regulatory role of glutaredoxins
Abstract
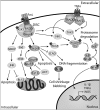
Significance: Glutaredoxin (Grx) is the primary enzyme responsible for catalysis of deglutathionylation of protein-mixed disulfides with glutathione (GSH) (protein-SSG). This reversible post-translational modification alters the activity and function of many proteins important in regulation of critical cellular processes. Aberrant regulation of protein glutathionylation/deglutathionylation reactions due to changes in Grx activity can disrupt both apoptotic and survival signaling pathways.
Recent advances: Grx is known to regulate the activity of many proteins through reversible glutathionylation, such as Ras, Fas, ASK1, NFκB, and procaspase-3, all of which play important roles in control of apoptosis. Reactive oxygen species and/or reactive nitrogen species mediate oxidative modifications of critical Cys residues on these apoptotic mediators, facilitating protein-SSG formation and thereby altering protein function and apoptotic signaling.
Critical issues: Much of what is known about the regulation of apoptotic mediators by Grx and reversible glutathionylation has been gleaned from in vitro studies of discrete apoptotic pathways. To relate these results to events in vivo it is important to examine changes in protein-SSG status in situ under natural cellular conditions, maintaining relevant GSH:GSSG ratios and using appropriate inducers of apoptosis.
Future directions: Apoptosis is a highly complex, tightly regulated process involving many different checks and balances. The influence of Grx activity on the interconnectivity among these various pathways remains unknown. Knowledge of the effects of Grx is essential for developing novel therapeutic approaches for treating diseases involving dysregulated apoptosis, such as cancer, heart disease, diabetes, and neurodegenerative diseases, where alterations in redox homeostasis are hallmarks for pathogenesis.
Figures






References
-
- Adachi T. Pimentel DR. Heibeck T. Hou X. Lee YJ. Jiang B. Ido Y. Cohen RA. S-glutathiolation of Ras mediates redox-sensitive signaling by angiotensin II in vascular smooth muscle cells. J Biol Chem. 2004;279:29857–29862. - PubMed
-
- Andersen JK. Oxidative stress in neurodegeneration: cause or consequence? Nat Med. 2004;10(Suppl):S18–S25. - PubMed
-
- Applegate MA. Humphries KM. Szweda LI. Reversible inhibition of alpha-ketoglutarate dehydrogenase by hydrogen peroxide: glutathionylation and protection of lipoic acid. Biochemistry. 2008;47:473–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

