Poly(ADP-ribose) regulates post-transcriptional gene regulation in the cytoplasm
- PMID: 22531498
- PMCID: PMC3495734
- DOI: 10.4161/rna.19899
Poly(ADP-ribose) regulates post-transcriptional gene regulation in the cytoplasm
Abstract
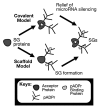
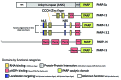
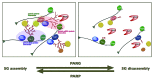
Since its discovery in 1963, poly(ADP-ribose) (pADPr) has been shown to play important functions in the nucleus of multicellular eukaryotes. Each of these functions centers upon DNA metabolism, including DNA-damage repair, chromatin remodeling, transcription and telomere functions. We recently described two novel functions for pADPr in the cytoplasm, both of which involve RNA metabolism - 1) the assembly of cytoplasmic stress granules, cellular macrostructures that aggregate translationally stalled mRNA/protein complexes, and 2) modulation of microRNA activities. Multiple stress granule-localized, post-transcriptional gene regulators, including microRNA-binding argonaute family members, are substrates for pADPr modification and are increasingly modified by pADPr upon stress. Interestingly, the cytoplasmic RNA regulatory functions for PARPs are likely mediated through activities of catalytically inactive PARP-13/ARTD13/ZC3HAV1/ZAP and mono/poly(ADP-ribose)-synthesizing enzymes, including PARP-5a/ARTD5/TNKS1, PARP-12/ARTD12/ZC3HDC1 and PARP-15/ARTD7/BAL3. These data are consistent with other recent work, which suggests that mono(ADP-ribosyl)ated residues can be poly(ADP-ribosyl)ated by different enzymes.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
