Modifying three-dimensional scaffolds from novel nanocomposite materials using dissolvable porogen particles for use in liver tissue engineering
- PMID: 22532408
- PMCID: PMC4107826
- DOI: 10.1177/0885328212445404
Modifying three-dimensional scaffolds from novel nanocomposite materials using dissolvable porogen particles for use in liver tissue engineering
Abstract
Background: Although hepatocytes have a remarkable regenerative power, the rapidity of acute liver failure makes liver transplantation the only definitive treatment. Attempts to incorporate engineered three-dimensional liver tissue in bioartificial liver devices or in implantable tissue constructs, to treat or bridge patients to self-recovery, were met with many challenges, amongst which is to find suitable polymeric matrices. We studied the feasibility of utilising nanocomposite polymers in three-dimensional scaffolds for hepatocytes.
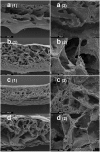
Materials and methods: Hepatocytes (HepG2) were seeded on a flat sheet and in three-dimensional scaffolds made of a nanocomposite polymer (Polyhedral Oligomeric Silsesquioxane [POSS]-modified polycaprolactone urea urethane) alone as well as with porogen particles, i.e. glucose, sodium bicarbonate and sodium chloride. The scaffold architecture, cell attachment and morphology were studied with scanning electron microscopy, and we assessed cell viability and functionality.
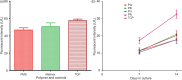
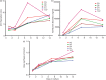
Results: Cell attachment to the scaffolds was demonstrated. The scaffold made with glucose particles as porogen showed a narrower range of pore size with higher porosity and better inter-pore communications and seemed to encourage near normal cell morphology. There was a steady increase of albumin secretion throughout the experiment while the control (monolayer cell culture) showed a steep decrease after day 7. At the end of the experiment, there was no significant difference in viability and functionality between the scaffolds and the control.
Conclusion: In this initial study, porogen particles were used to modify the scaffolds produced from the novel polymer. Although there was no significance against the control in functionality and viability, the demonstrable attachment on scanning electron microscopy suggest potential roles for this polymer and in particular for scaffolds made with glucose particles in liver tissue engineering.
Keywords: HepG2; POSS-modified polycaprolactone urea urethane; hepatocytes; nanocomposite polymers; porogens; scaffolds; tissue engineering.
Figures




Similar articles
-
Recent Advances in Polyurethane/POSS Hybrids for Biomedical Applications.Molecules. 2021 Dec 22;27(1):40. doi: 10.3390/molecules27010040. Molecules. 2021. PMID: 35011280 Free PMC article. Review.
-
A new biodegradable nanocomposite based on polyhedral oligomeric silsesquioxane nanocages: cytocompatibility and investigation into electrohydrodynamic jet fabrication techniques for tissue-engineered scaffolds.Biotechnol Appl Biochem. 2009 Jan;52(Pt 1):1-8. doi: 10.1042/BA20070256. Biotechnol Appl Biochem. 2009. PMID: 18402554
-
In vitro small intestinal epithelial cell growth on a nanocomposite polycaprolactone scaffold.Biotechnol Appl Biochem. 2009 Dec 4;54(4):221-9. doi: 10.1042/BA20090214. Biotechnol Appl Biochem. 2009. PMID: 19860739 Free PMC article.
-
Surface modification of a POSS-nanocomposite material to enhance cellular integration of a synthetic bioscaffold.Biomaterials. 2016 Mar;83:283-93. doi: 10.1016/j.biomaterials.2016.01.005. Epub 2016 Jan 5. Biomaterials. 2016. PMID: 26790147 Free PMC article.
-
Recent trends in the application of widely used natural and synthetic polymer nanocomposites in bone tissue regeneration.Mater Sci Eng C Mater Biol Appl. 2020 May;110:110698. doi: 10.1016/j.msec.2020.110698. Epub 2020 Jan 29. Mater Sci Eng C Mater Biol Appl. 2020. PMID: 32204012 Free PMC article. Review.
Cited by
-
First Application of a Mixed Porcine-Human Repopulated Bioengineered Liver in a Preclinical Model of Post-Resection Liver Failure.Biomedicines. 2024 Jun 7;12(6):1272. doi: 10.3390/biomedicines12061272. Biomedicines. 2024. PMID: 38927479 Free PMC article.
-
Recent Advances in Polyurethane/POSS Hybrids for Biomedical Applications.Molecules. 2021 Dec 22;27(1):40. doi: 10.3390/molecules27010040. Molecules. 2021. PMID: 35011280 Free PMC article. Review.
References
-
- Facts about liver disease, http://www.britishlivertrust.org. uk/data/5/pages/2 aspx?pid=280 (2008). (accessed 31 January 2008)
-
- Standards for solid organ transplantation in the United Kingdom, http://www.bts.org.uk/Forms/standards%20document%20edition%202%20-%20final pdf (2003 July). (accessed 31 January 2008)
-
- Transplant activity in the UK, http://www.uktransplant. org.uk/ukt/statistics/transplant_activity_report/current_activity_report... pdf (2007 August). (accessed 31 January 2008)
-
- Vienken J, Christmann H. How can liver toxins be removed? Filtration and adsorption with the Prometheus system. Ther Apher Dial 2006; 10(2): 125–131 - PubMed
-
- Laleman W, Wilmer A, Evenepoel P, et al. Review article: non-biological liver support in liver failure. Aliment Pharmacol Ther 2006; 23(3): 351–363 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

