The effects of NMDA subunit composition on calcium influx and spike timing-dependent plasticity in striatal medium spiny neurons
- PMID: 22536151
- PMCID: PMC3334887
- DOI: 10.1371/journal.pcbi.1002493
The effects of NMDA subunit composition on calcium influx and spike timing-dependent plasticity in striatal medium spiny neurons
Abstract
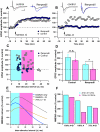
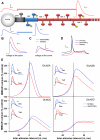
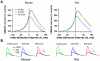
Calcium through NMDA receptors (NMDARs) is necessary for the long-term potentiation (LTP) of synaptic strength; however, NMDARs differ in several properties that can influence the amount of calcium influx into the spine. These properties, such as sensitivity to magnesium block and conductance decay kinetics, change the receptor's response to spike timing dependent plasticity (STDP) protocols, and thereby shape synaptic integration and information processing. This study investigates the role of GluN2 subunit differences on spine calcium concentration during several STDP protocols in a model of a striatal medium spiny projection neuron (MSPN). The multi-compartment, multi-channel model exhibits firing frequency, spike width, and latency to first spike similar to current clamp data from mouse dorsal striatum MSPN. We find that NMDAR-mediated calcium is dependent on GluN2 subunit type, action potential timing, duration of somatic depolarization, and number of action potentials. Furthermore, the model demonstrates that in MSPNs, GluN2A and GluN2B control which STDP intervals allow for substantial calcium elevation in spines. The model predicts that blocking GluN2B subunits would modulate the range of intervals that cause long term potentiation. We confirmed this prediction experimentally, demonstrating that blocking GluN2B in the striatum, narrows the range of STDP intervals that cause long term potentiation. This ability of the GluN2 subunit to modulate the shape of the STDP curve could underlie the role that GluN2 subunits play in learning and development.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



References
-
- Yin HH, Mulcare SP, Hilário MRF, Clouse E, Holloway T, et al. Dynamic reorganization of striatal circuits during the acquisition and consolidation of a skill. Nat Neurosci. 2009;12:333–341. doi: 10.1038/nn.2261. - DOI - PMC - PubMed
-
- Paillé V, Picconi B, Bagetta V, Ghiglieri V, Sgobio C, et al. Distinct levels of dopamine denervation differentially alter striatal synaptic plasticity and NMDA receptor subunit composition. J Neurosci. 2010;30:14182–14193. doi: 10.1523/JNEUROSCI.2149-10.2010. - DOI - PMC - PubMed
-
- Kung VWS, Hassam R, Morton AJ, Jones S. Dopamine-dependent long term potentiation in the dorsal striatum is reduced in the R6/2 mouse model of Huntington's disease. Neuroscience. 2007;146:1571–1580. doi: 10.1016/j.neuroscience.2007.03.036. - DOI - PubMed
-
- Bender VA, Bender KJ, Brasier DJ, Feldman DE. Two coincidence detectors for spike timing-dependent plasticity in somatosensory cortex. J Neurosci. 2006;26:4166–4177. doi: 10.1523/JNEUROSCI.0176-06.2006. - DOI - PMC - PubMed
-
- Shindou T, Ochi-Shindou M, Wickens JR. A Ca(2+) threshold for induction of spike-timing-dependent depression in the mouse striatum. J Neurosci. 2011;31:13015–13022. doi: 10.1523/JNEUROSCI.3206-11.2011. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

