Vitamin D deficiency induces cardiac hypertrophy and inflammation in epicardial adipose tissue in hypercholesterolemic swine
- PMID: 22537546
- PMCID: PMC3411274
- DOI: 10.1016/j.yexmp.2012.04.006
Vitamin D deficiency induces cardiac hypertrophy and inflammation in epicardial adipose tissue in hypercholesterolemic swine
Abstract
Introduction: Vitamin D is a sectosteroid that functions through Vitamin D receptor (VDR), a transcription factor, which controls the transcription of many targets genes. Vitamin D deficiency has been linked with cardiovascular diseases, including heart failure and coronary artery disease. Suppressor of cytokine signaling (SOCS)3 regulates different biological processes such as inflammation and cellular differentiation and is an endogenous negative regulator of cardiac hypertrophy.
Objective: The purpose of this study was to test the hypothesis that vitamin D deficiency causes cardiomyocyte hypertrophy and increased proinflammatory profile in epicardial adipose tissue (EAT), and this correlates with decreased expression of SOCS3 in cardiomyocytes and EAT.
Methods: Eight female Yucatan miniswine were fed vitamin D-sufficient (900 IU/d) or vitamin D-deficient hypercholesterolemic diet. Lipid profile, metabolic panel, and serum 25(OH)D levels were regularly measured. After 12 months animals were euthanized and histological, immunohistochemical and qPCR studies were performed on myocardium and epicardial fat.
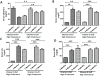
Results: Histological studies showed cardiac hypertrophy, as judged by cardiac myocyte cross sectional area, in the vitamin D-deficient group. Immunohistochemical and qPCR analyses showed significantly decreased mRNA and protein expression of VDR and SOCS3 in cardiomyocytes of vitamin D-deficient animals. EAT from vitamin D-deficient group had significantly higher expression of TNF-α, IL-6, MCP-1, and decreased adiponectin in association with increased inflammatory cellular infiltrate. Interestingly, EAT from vitamin D-deficient group had significantly decreased expression of SOCS3.
Conclusion: These data suggest that vitamin D deficiency induces hypertrophy in cardiomyocytes which is associated with decreased expression of VDR and SOCS3. Vitamin D deficiency is also associated with increased inflammatory markers in EAT. Activity of VDR in the body is controlled through regulation of vitamin D metabolites. Therefore, restoration of VDR function by supplementation of VDR ligands in vitamin D-deficient population might be helpful in reducing inflammation and cardiovascular risk.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures






References
-
- Antoniades C, Antonopoulos AS, Tousoulis D, Stefanadis C. Adiponectin: from obesity to cardiovascular disease. Obes Rev. 2009;10:269–279. - PubMed
-
- Chen S, Wu J, Hsieh JC, Whitfield GK, Jurutka PW, Haussler MR, Gardner DG. Suppression of ANP gene transcription by liganded vitamin D receptor: involvement of specific receptor domains. Hypertension. 1998;31:1338–1342. - PubMed
-
- Dobnig H, Pilz S, Scharnagl H, Renner W, Seelhorst U, Wellnitz B, Kinkeldei J, Boehm BO, Weihrauch G, Maerz W. Independent association of low serum 25-hydroxyvitamin d and 1,25-dihydroxyvitamin d levels with all-cause and cardiovascular mortality. Arch Intern Med. 2008;168:1340–1349. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

