An interspecific Nicotiana hybrid as a useful and cost-effective platform for production of animal vaccines
- PMID: 22539991
- PMCID: PMC3334924
- DOI: 10.1371/journal.pone.0035688
An interspecific Nicotiana hybrid as a useful and cost-effective platform for production of animal vaccines
Abstract
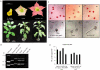
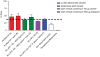
The use of transgenic plants to produce novel products has great biotechnological potential as the relatively inexpensive inputs of light, water, and nutrients are utilised in return for potentially valuable bioactive metabolites, diagnostic proteins and vaccines. Extensive research is ongoing in this area internationally with the aim of producing plant-made vaccines of importance for both animals and humans. Vaccine purification is generally regarded as being integral to the preparation of safe and effective vaccines for use in humans. However, the use of crude plant extracts for animal immunisation may enable plant-made vaccines to become a cost-effective and efficacious approach to safely immunise large numbers of farm animals against diseases such as avian influenza. Since the technology associated with genetic transformation and large-scale propagation is very well established in Nicotiana, the genus has attributes well-suited for the production of plant-made vaccines. However the presence of potentially toxic alkaloids in Nicotiana extracts impedes their use as crude vaccine preparations. In the current study we describe a Nicotiana tabacum and N. glauca hybrid that expresses the HA glycoprotein of influenza A in its leaves but does not synthesize alkaloids. We demonstrate that injection with crude leaf extracts from these interspecific hybrid plants is a safe and effective approach for immunising mice. Moreover, this antigen-producing alkaloid-free, transgenic interspecific hybrid is vigorous, with a high capacity for vegetative shoot regeneration after harvesting. These plants are easily propagated by vegetative cuttings and have the added benefit of not producing viable pollen, thus reducing potential problems associated with bio-containment. Hence, these Nicotiana hybrids provide an advantageous production platform for partially purified, plant-made vaccines which may be particularly well suited for use in veterinary immunization programs.
Conflict of interest statement
Figures




References
-
- Walmsley AM, Arntzen CJ. Plants for delivery of edible vaccines. Current Opinion in Biotechnology. 2000;11:126–129. - PubMed
-
- Rigano MM, Manna C, Giulini A, Vitale A, Cardi T. Plants as biofactories for the production of subunit vaccines against bio-security-related bacteria and viruses. Vaccine. 2009;27:3463–3466. - PubMed
-
- D'Aoust MA, Couture MM, Charland N, Trepanier S, Landry N, et al. The production of hemagglutinin-based virus-like particles in plants: a rapid, efficient and safe response to pandemic influenza. Plant Biotechnol J. 2010;8:607–619. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

