Activation of the renal Na+:Cl- cotransporter by angiotensin II is a WNK4-dependent process
- PMID: 22550170
- PMCID: PMC3356635
- DOI: 10.1073/pnas.1200947109
Activation of the renal Na+:Cl- cotransporter by angiotensin II is a WNK4-dependent process
Abstract
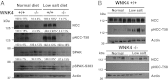
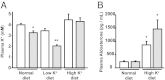
Pseudohypoaldosteronism type II is a salt-sensitive form of hypertension with hyperkalemia in humans caused by mutations in the with-no-lysine kinase 4 (WNK4). Several studies have shown that WNK4 modulates the activity of the renal Na(+)Cl(-) cotransporter, NCC. Because the renal consequences of WNK4 carrying pseudoaldosteronism type II mutations resemble the response to intravascular volume depletion (promotion of salt reabsorption without K(+) secretion), a condition that is associated with high angiotensin II (AngII) levels, it has been proposed that AngII signaling might affect WNK4 modulation of the NCC. In Xenopus laevis oocytes, WNK4 is required for modulation of NCC activity by AngII. To demonstrate that WNK4 is required in the AngII-mediated regulation of NCC in vivo, we used a total WNK4-knockout mouse strain (WNK4(-/-)). WNK4 mRNA and protein expression were absent in WNK4(-/-) mice, which exhibited a mild Gitelman-like syndrome, with normal blood pressure, increased plasma renin activity, and reduced NCC expression and phosphorylation at T-58. Immunohistochemistry revealed normal morphology of the distal convoluted tubule with reduced NCC expression. Low-salt diet or infusion of AngII for 4 d induced phosphorylation of STE20/SPS1-related proline/alanine-rich kinase (SPAK) and of NCC at S-383 and T-58, respectively, in WNK4(+/+) but not WNK4(-/-) mice. Thus, the absence of WNK4 in vivo precludes NCC and SPAK phosphorylation promoted by a low-salt diet or AngII infusion, suggesting that AngII action on the NCC occurs via a WNK4-SPAK-dependent signaling pathway. Additionally, stimulation of aldosterone secretion by AngII, but not by a high-K(+) diet, was impaired in WNK4(-/-) mice.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Lifton RP, Gharavi AG, Geller DS. Molecular mechanisms of human hypertension. Cell. 2001;104:545–556. - PubMed
-
- Mayan H, et al. Pseudohypoaldosteronism type II: Marked sensitivity to thiazides, hypercalciuria, normomagnesemia, and low bone mineral density. J Clin Endocrinol Metab. 2002;87:3248–3254. - PubMed
-
- Simon DB, et al. Gitelman’s variant of Bartter’s syndrome, inherited hypokalaemic alkalosis, is caused by mutations in the thiazide-sensitive Na-Cl cotransporter. Nat Genet. 1996;12:24–30. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

