Benznidazole-resistance in Trypanosoma cruzi is a readily acquired trait that can arise independently in a single population
- PMID: 22551809
- PMCID: PMC3379838
- DOI: 10.1093/infdis/jis331
Benznidazole-resistance in Trypanosoma cruzi is a readily acquired trait that can arise independently in a single population
Abstract
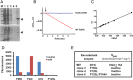
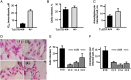
Benznidazole is the frontline drug used against Trypanosoma cruzi, the causative agent of Chagas disease. However, treatment failures are often reported. Here, we demonstrate that independently acquired mutations in the gene encoding a mitochondrial nitroreductase (TcNTR) can give rise to distinct drug-resistant clones within a single population. Following selection of benznidazole-resistant parasites, all clones examined had lost one of the chromosomes containing the TcNTR gene. Sequence analysis of the remaining TcNTR allele revealed 3 distinct mutant genes in different resistant clones. Expression studies showed that these mutant proteins were unable to activate benznidazole. This correlated with loss of flavin mononucleotide binding. The drug-resistant phenotype could be reversed by transfection with wild-type TcNTR. These results identify TcNTR as a central player in acquired resistance to benznidazole. They also demonstrate that T. cruzi has a propensity to undergo genetic changes that can lead to drug resistance, a finding that has implications for future therapeutic strategies.
Figures


References
-
- Moncayo A, Silveira AC. Current epidemiological trends for Chagas disease in Latin America and future challenges in epidemiology, surveillance and health policy. Mem Inst Oswaldo Cruz. 2009;104:17–30. - PubMed
-
- Bern C, Montgomery SP. An estimate of the burden of Chagas disease in the United States. Clin Infect Dis. 2009;49:e52–4. - PubMed
-
- Wilkinson SR, Kelly JM. Trypanocidal drugs: mechanisms, resistance and new targets. Expert Rev Mol Med. 2009;11:1–24. - PubMed
-
- Castro JA, de Mecca MM, Bartel LC. Toxic side effects of drugs used to treat Chagas’ disease (American trypanosomiasis) Hum Exp Toxicol. 2006;25:471–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

