The role and source of tumor necrosis factor-α in hemorrhage-induced priming for septic lung injury
- PMID: 22552013
- PMCID: PMC3356491
- DOI: 10.1097/SHK.0b013e318254fa6a
The role and source of tumor necrosis factor-α in hemorrhage-induced priming for septic lung injury
Abstract
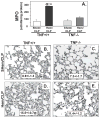
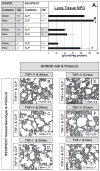
Tumor necrosis factor α (TNF-α) has been reported to be a key component of the functional priming, of both myeloid and nonmyeloid cells, that is thought to contribute to the lung's increased susceptibility to injury following shock. Not surprisingly, we found that mice deficient in TNF-α exhibited reduced acute lung injury (ALI) resultant from the combined insults of hemorrhagic shock and sepsis. However, we found that when we adoptively transferred neutrophils from mice expressing TNF-α to neutrophil-depleted mice that lacked TNF-α, they were not able to serve as priming stimulus for the development of ALI. Based on these findings, we proposed that resident lung tissue cells mediate TNF-α priming. To begin to unravel the complex signaling pathway of various resident lung tissue cells in TNF-α-induced priming, we compared the effect of local (intratracheal [i.t.]) versus systemic [intravenous (i.v.)] delivery of TNF-α small interference (siRNA). We hypothesized that alternately suppressing expression of TNF-α in lung endothelial (i.v.) or epithelial (i.t.) cells would produce a differential effect in shock-induced ALI. We found that when in vivo siRNA i.t. or i.v. against TNF-α was administered to C57/BL6 mice at 2 h after hemorrhage, 24 h before septic challenge, that systemic/i.v., but not i.t., delivery of TNF-α siRNA following hemorrhage priming significantly reduces expression of indices of ALI compared with controls. These findings suggest that an absence of local lung tissue TNF-α significantly reduces lung tissue injury following hemorrhage priming for ALI and that pulmonary endothelial and/or other possible vascular resident cells, not epithelial cells, play a greater role in mediating the TNF-α priming response in a mouse model of hemorrhage/sepsis-induced ALI.
Conflict of interest statement
Authors have no existing conflicts of interest related to the subject matter of this manuscript.
Figures


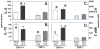


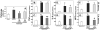

References
-
- Abraham E, Carmody A, Shenkar R, Arcaroli J. Neutrophils as early immunologic effectors in hemorrhage- or endotoxemia-induced acute lung injury. Am J Physiol Lung Cell Mol Physiol. 2000;279:L1137–L1145. - PubMed
-
- Alarcon P, Conejeros I, Carretta MD, Concha C, Jara E, Tadich N, Hidalgo MA, Burgos RA. D-Lactic acid interferes with the effects of platelet activating factor on bovine neutrophils. Veterinary Immun and Immunopath. 2011;144:68–78. - PubMed
-
- Aleku M, Fisch G, Mopert K, Keil O, Woflgang A, Kaufmann J, Santel A. Intracellular localization of lipoplexed siRNA in vascular endothelial cells of different mouse tissues. Microvasc Res. 2008;76:31–41. - PubMed
-
- Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med. 2001;29:1303–1310. - PubMed
-
- Ayala A, Chung CS, Lomas J, Grutkoski PS, Doughty LA, Simms HH. A mouse model of priming for acute lung injury following shock. Shock. 2001;15:83S.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

