Smoothened antagonists reverse taxane resistance in ovarian cancer
- PMID: 22553355
- PMCID: PMC3392529
- DOI: 10.1158/1535-7163.MCT-11-1058
Smoothened antagonists reverse taxane resistance in ovarian cancer
Abstract
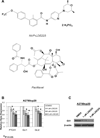
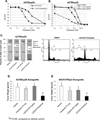
The hedgehog pathway has been implicated in the formation and maintenance of a variety of malignancies, including ovarian cancer; however, it is unknown whether hedgehog signaling is involved in ovarian cancer chemoresistance. The goal of this study was to determine the effects of antagonizing the hedgehog receptor, Smoothened (Smo), on chemotherapy response in ovarian cancer. Expression of hedgehog pathway members was assessed in three pairs of parental and chemotherapy-resistant ovarian cancer cell lines (A2780ip2/A2780cp20, SKOV3ip1/SKOV3TRip2, HeyA8/HeyA8MDR) using quantitative PCR and Western blot analysis. Cell lines were exposed to increasing concentrations of two different Smo antagonists (cyclopamine, LDE225) alone and in combination with carboplatin or paclitaxel. Selective knockdown of Smo, Gli1, or Gli2 was achieved using siRNA constructs. Cell viability was assessed by MTT assay. A2780cp20 and SKOV3TRip2 orthotopic xenografts were treated with vehicle, LDE225, paclitaxel, or combination therapy. Chemoresistant cell lines showed higher expression (>2-fold, P < 0.05) of hedgehog signaling components compared with their respective parental lines. Smo antagonists sensitized chemotherapy-resistant cell lines to paclitaxel, but not to carboplatin. LDE225 treatment also increased sensitivity of ALDH-positive cells to paclitaxel. A2780cp20 and SKOV3TRip2 xenografts treated with combined LDE225 and paclitaxel had significantly less tumor burden than those treated with vehicle or either agent alone. Increased taxane sensitivity seems to be mediated by a decrease in P-glycoprotein (MDR1) expression. Selective knockdown of Smo, Gli1, or Gli2 all increased taxane sensitivity. Smo antagonists reverse taxane resistance in chemoresistant ovarian cancer models, suggesting combined anti-hedgehog and chemotherapies could provide a useful therapeutic strategy for ovarian cancer.
©2012 AACR.
Conflict of interest statement
Figures



References
-
- Bhoola S, Hoskins WJ. Diagnosis and management of epithelial ovarian cancer. Obstet Gynecol. 2006;107:1399–1410. - PubMed
-
- Hooper JE, Scott MP. Communicating with Hedgehogs. Nature reviews. 2005;6:306–317. - PubMed
-
- Pasca di Magliano M, Hebrok M. Hedgehog signalling in cancer formation and maintenance. Nat Rev Cancer. 2003;3:903–911. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

