Potential state-selective hydrogen bond formation can modulate activation and desensitization of the α7 nicotinic acetylcholine receptor
- PMID: 22556416
- PMCID: PMC3381156
- DOI: 10.1074/jbc.M112.339796
Potential state-selective hydrogen bond formation can modulate activation and desensitization of the α7 nicotinic acetylcholine receptor
Abstract
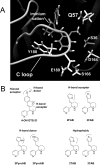
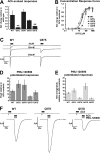
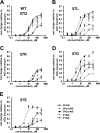
A series of arylidene anabaseines were synthesized to probe the functional impact of hydrogen bonding on human α7 nicotinic acetylcholine receptor (nAChR) activation and desensitization. The aryl groups were either hydrogen bond acceptors (furans), donors (pyrroles), or neither (thiophenes). These compounds were tested against a series of point mutants of the ligand-binding domain residue Gln-57, a residue hypothesized to be proximate to the aryl group of the bound agonist and a putative hydrogen bonding partner. Q57K, Q57D, Q57E, and Q57L were chosen to remove the dual hydrogen bonding donor/acceptor ability of Gln-57 and replace it with hydrogen bond donating, hydrogen bond accepting, or nonhydrogen bonding ability. Activation of the receptor was compromised with hydrogen bonding mismatches, for example, pairing a pyrrole with Q57K or Q57L, or a furan anabaseine with Q57D or Q57E. Ligand co-applications with the positive allosteric modulator PNU-120596 produced significantly enhanced currents whose degree of enhancement was greater for 2-furans or -pyrroles than for their 3-substituted isomers, whereas the nonhydrogen bonding thiophenes failed to show this correlation. Interestingly, the PNU-120596 agonist co-application data revealed that for wild-type α7 nAChR, the 3-furan desensitized state was relatively stabilized compared with that of 2-furan, a reversal of the relationship observed with respect to the barrier for entry into the desensitized state. These data highlight the importance of hydrogen bonding on the receptor-ligand state, and suggest that it may be possible to fine-tune features of agonists that mediate state selection in the nAChR.
Figures




Similar articles
-
Point-to-point ligand-receptor interactions across the subunit interface modulate the induction and stabilization of conformational states of alpha7 nAChR by benzylidene anabaseines.Biochem Pharmacol. 2013 Mar 15;85(6):817-28. doi: 10.1016/j.bcp.2013.01.010. Epub 2013 Jan 23. Biochem Pharmacol. 2013. PMID: 23352650 Free PMC article.
-
Docking studies of benzylidene anabaseine interactions with α7 nicotinic acetylcholine receptor (nAChR) and acetylcholine binding proteins (AChBPs): application to the design of related α7 selective ligands.Eur J Med Chem. 2011 Nov;46(11):5625-35. doi: 10.1016/j.ejmech.2011.09.033. Epub 2011 Sep 29. Eur J Med Chem. 2011. PMID: 21986237 Free PMC article.
-
Activation and desensitization of nicotinic alpha7-type acetylcholine receptors by benzylidene anabaseines and nicotine.J Pharmacol Exp Ther. 2009 May;329(2):791-807. doi: 10.1124/jpet.108.150151. Epub 2009 Feb 17. J Pharmacol Exp Ther. 2009. PMID: 19223664 Free PMC article.
-
Nicotinic receptors containing the alpha7 subunit: a model for rational drug design.Curr Med Chem. 2008;15(28):2921-32. doi: 10.2174/092986708786848703. Curr Med Chem. 2008. PMID: 19075644 Free PMC article. Review.
-
Alzheimer's drug design based upon an invertebrate toxin (anabaseine) which is a potent nicotinic receptor agonist.Invert Neurosci. 1997 Sep-Dec;3(2-3):251-9. doi: 10.1007/BF02480382. Invert Neurosci. 1997. PMID: 9783447 Review.
Cited by
-
An outline of desensitization in pentameric ligand-gated ion channel receptors.Cell Mol Life Sci. 2013 Apr;70(7):1241-53. doi: 10.1007/s00018-012-1133-z. Epub 2012 Aug 31. Cell Mol Life Sci. 2013. PMID: 22936353 Free PMC article. Review.
-
Analysis of the stability and affinity of BlaR-CTD protein to β-lactam antibiotics based on docking and mutagenesis studies.J Biol Eng. 2019 Mar 29;13:27. doi: 10.1186/s13036-019-0157-4. eCollection 2019. J Biol Eng. 2019. PMID: 30976316 Free PMC article.
-
Discovery of an intrasubunit nicotinic acetylcholine receptor-binding site for the positive allosteric modulator Br-PBTC.J Biol Chem. 2019 Aug 9;294(32):12132-12145. doi: 10.1074/jbc.RA118.006253. Epub 2019 Jun 20. J Biol Chem. 2019. PMID: 31221718 Free PMC article.
-
Persistent activation of α7 nicotinic ACh receptors associated with stable induction of different desensitized states.Br J Pharmacol. 2018 Jun;175(11):1838-1854. doi: 10.1111/bph.13851. Epub 2017 Jun 8. Br J Pharmacol. 2018. PMID: 28477386 Free PMC article.
-
Impact of modulation of the α7 nicotinic acetylcholine receptor on nicotine reward in the mouse conditioned place preference test.Psychopharmacology (Berl). 2019 Dec;236(12):3593-3599. doi: 10.1007/s00213-019-05331-y. Epub 2019 Jul 13. Psychopharmacology (Berl). 2019. PMID: 31302720 Free PMC article.
References
-
- Millar N. S., Gotti C. (2009) Diversity of vertebrate nicotinic acetylcholine receptors. Neuropharmacology 56, 237–246 - PubMed
-
- Dani J. A., Radcliffe K. A., Pidoplichko V. I. (2000) Variations in desensitization of nicotinic acetylcholine receptors from hippocampus and midbrain dopamine areas. Eur. J. Pharmacol. 393, 31–38 - PubMed
-
- Quick M. W., Lester R. A. (2002) Desensitization of neuronal nicotinic receptors. J. Neurobiol. 53, 457–478 - PubMed
-
- Le Novère N., Corringer P. J., Changeux J. P. (2002) The diversity of subunit composition in nAChRs. Evolutionary origins, physiologic and pharmacologic consequences. J. Neurobiol. 53, 447–456 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

