JKA97, a novel benzylidene analog of harmine, exerts anti-cancer effects by inducing G1 arrest, apoptosis, and p53-independent up-regulation of p21
- PMID: 22558087
- PMCID: PMC3338851
- DOI: 10.1371/journal.pone.0034303
JKA97, a novel benzylidene analog of harmine, exerts anti-cancer effects by inducing G1 arrest, apoptosis, and p53-independent up-regulation of p21
Abstract
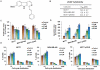
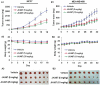
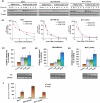
JKA97, a benzylidene analog of harmine, has been found to be a promising drug candidate for human cancer therapy, although the underlying molecular mechanisms have not been fully demonstrated. In this study, we evaluated the effects of JKA97 on human breast cancer cells in vitro and in vivo. JKA97 inhibited the growth and proliferation of MCF7 (p53 wild-type), MCF7 (p53 knockdown), and MDA-MB-468 (p53 mutant) cells in a dose-dependent manner. Treatment with JKA97 arrested breast cancer cells in G1 phase and induced apoptosis. JKA97 also significantly suppressed the growth of MCF7 and MDA-MB-468 xenograft tumors. It regulated the expression levels of G1 phase regulators, such as p21, p27, cyclinE, and cylinD1. JKA97 activated p21 transcription, independent of p53, but had little effect on p21 protein stability/degradation. In summary, our results suggest that JKA97 inhibits human breast cancer cell growth through activating p21, independent of p53, which provides a basis for developing this compound as a novel drug for human breast cancer therapy.
Conflict of interest statement
Figures

References
-
- Jemal A, Bray F, Center MM, Ferlay J, Ward E, et al. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90. - PubMed
-
- Nathanson KL, Domchek SM. Therapeutic approaches for women predisposed to breast cancer. Annu Rev Med. 2011;62:295–306. - PubMed
-
- Oakman C, Santarpia L, Di Leo A. Breast cancer assessment tools and optimizing adjuvant therapy. Nat Rev Clin Oncol. 2010;7:725–732. - PubMed
-
- Eskelinen M, Hippelainen M, Kettunen J, Salmela E, Penttila I, et al. Clinical value of serum tumour markers TPA, TPS, TAG 12, CA 15-3 and MCA in breast cancer diagnosis; results from a prospective study. Anticancer Res. 1994;14:699–703. - PubMed
-
- Koehn FE, Carter GT. The evolving role of natural products in drug discovery. Nat Rev Drug Discov. 2005;4:206–220. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

