Muscle-specific deletion of carnitine acetyltransferase compromises glucose tolerance and metabolic flexibility
- PMID: 22560225
- PMCID: PMC3348515
- DOI: 10.1016/j.cmet.2012.04.005
Muscle-specific deletion of carnitine acetyltransferase compromises glucose tolerance and metabolic flexibility
Abstract
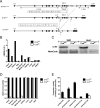
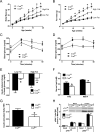
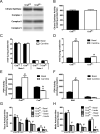
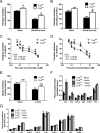
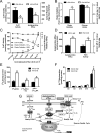
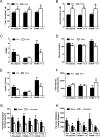
The concept of "metabolic inflexibility" was first introduced to describe the failure of insulin-resistant human subjects to appropriately adjust mitochondrial fuel selection in response to nutritional cues. This phenomenon has since gained increasing recognition as a core component of the metabolic syndrome, but the underlying mechanisms have remained elusive. Here, we identify an essential role for the mitochondrial matrix enzyme, carnitine acetyltransferase (CrAT), in regulating substrate switching and glucose tolerance. By converting acetyl-CoA to its membrane permeant acetylcarnitine ester, CrAT regulates mitochondrial and intracellular carbon trafficking. Studies in muscle-specific Crat knockout mice, primary human skeletal myocytes, and human subjects undergoing L-carnitine supplementation support a model wherein CrAT combats nutrient stress, promotes metabolic flexibility, and enhances insulin action by permitting mitochondrial efflux of excess acetyl moieties that otherwise inhibit key regulatory enzymes such as pyruvate dehydrogenase. These findings offer therapeutically relevant insights into the molecular basis of metabolic inflexibility.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures
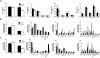
References
-
- Albarado DC, McClaine J, Stephens JM, Mynatt RL, Ye J, Bannon AW, Richards WG, Butler AA. Impaired coordination of nutrient intake and substrate oxidation in melanocortin-4 receptor knockout mice. Endocrinology. 2004;145:243–252. - PubMed
-
- An J, Muoio DM, Shiota M, Fujimoto Y, Cline GW, Shulman GI, Koves TR, Stevens R, Millington D, Newgard CB. Hepatic expression of malonyl-CoA decarboxylase reverses muscle, liver and whole-animal insulin resistance. NatMed. 2004;10:268–274. - PubMed
-
- Becker TC, BeltrandelRio H, Noel RJ, Johnson JH, Newgard CB. Overexpression of hexokinase I in isolated islets of Langerhans via recombinant adenovirus. Enhancement of glucose metabolism and insulin secretion at basal but not stimulatory glucose levels. J Biol Chem. 1994;269:21234–21238. - PubMed
-
- Beenakkers AM, Klingenberg M. Carnitine-Coenzyme a Transacetylase in Mitochondria from Various Organs. Biochim Biophys Acta. 1964;84:205–207. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

