Efficacy of a smoking quit line in the military: baseline design and analysis
- PMID: 22561390
- PMCID: PMC4521417
- DOI: 10.1016/j.cct.2012.04.009
Efficacy of a smoking quit line in the military: baseline design and analysis
Abstract
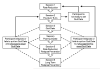
Thirty percent of all military personnel smoke cigarettes. Because of the negative health consequences and their impact on physical fitness, overall health, and military readiness, the Department of Defense has identified the reduction of tobacco use as a priority of US military forces. This study aims to evaluate the one-year efficacy of a proactive versus reactive smoking quit line in the US military with adjunctive nicotine replacement therapy (NRT) in both groups. This paper reports on the baseline variables of the first 1000 participants randomized, the design, and proposed analysis of the randomized two-arm clinical trial "Efficacy of a Tobacco Quit Line in the Military". Participants are adult smokers who are Armed Forces Active Duty personnel, retirees, Reservist, National Guard and family member healthcare beneficiaries. All participants are randomized to either the Counselor Initiated (proactive) group, receiving 6 counseling sessions in addition to an 8-week supply of NRT, or the Self-Paced (reactive) group, in which they may call the quit line themselves to receive the same counseling sessions, in addition to a 2-week supply of NRT. The primary outcome measure of the study is self-reported smoking abstinence at 1-year follow-up. Results from this study will be the first to provide evidence for the efficacy of an intensive Counselor Initiated quit line with provided NRT in military personnel and could lead to dissemination throughout the US Air Force, the armed forces population as a whole and ultimately to civilian personnel that do not have ready access to preventive health services.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures
References
-
- Centers for Disease Control and Prevention Vital signs: current cigarette smoking among adults aged ≥18 years — United States, 2009. Morb Mortal Wkly Rep. 2010;59:1135–40. - PubMed
-
- Centers for Disease Control and Prevention State-specific prevalence of cigarette smoking and smokeless tobacco use among adults— United States, 2009. Morb Mortal Wkly Rep. 2010;59:1400–6. - PubMed
-
- Centers for Disease Control and Prevention Current cigarette smoking among adults aged ≥18 years — United States, 2005-2010. MorbMortal Wkly Rep. 2011;60:1207–12. - PubMed
-
- Bray RM, Pemberton MR, Hourani LL, Witt M, Olmsted KL, Brown JM, et al. Department of Defense survey of health related behaviors among active duty military personnel. RTI International; Research Triangle Park, NC: 2008.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous