Increased late sodium current contributes to long QT-related arrhythmia susceptibility in female mice
- PMID: 22562703
- PMCID: PMC3633400
- DOI: 10.1093/cvr/cvs160
Increased late sodium current contributes to long QT-related arrhythmia susceptibility in female mice
Abstract
Aims: Female gender is a risk factor for long QT-related arrhythmias, but the underlying mechanisms remain uncertain. Here, we tested the hypothesis that gender-dependent function of the post-depolarization 'late' sodium current (I(Na-L)) contributes.
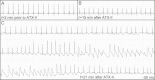
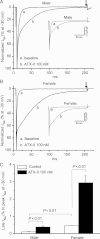
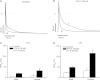
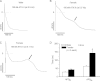
Methods and results: Studies were conducted in mice in which the canonical cardiac sodium channel Scn5a locus was disrupted, and expression of human wild-type SCN5A cDNA substituted. Baseline QT intervals were similar in male and female mice, but exposure to the sodium channel opener anemone toxin ATX-II elicited polymorphic ventricular tachycardia in 0/9 males vs. 6/9 females. Ventricular I(Na-L) and action potential durations were increased in myocytes isolated from female mice compared with those from males before and especially after treatment with ATX-II. Further, ATX-II elicited potentially arrhythmogenic early afterdepolarizations in myocytes from 0/5 male mice and 3/5 female mice.
Conclusion: These data identify variable late I(Na) as a modulator of gender-dependent arrhythmia susceptibility.
Figures

Comment in
-
Female gender: risk factor for congenital long QT-related arrhythmias.Cardiovasc Res. 2012 Aug 1;95(3):263-4. doi: 10.1093/cvr/cvs203. Epub 2012 Jun 21. Cardiovasc Res. 2012. PMID: 22721991 No abstract available.
References
-
- Schwartz PJ, Wolf S. QT interval prolongation as predictor of sudden death in patients with myocardial infarction. Circulation. 1978;57:1074–1077. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

