Zebrafish chemical screening reveals the impairment of dopaminergic neuronal survival by cardiac glycosides
- PMID: 22563390
- PMCID: PMC3338518
- DOI: 10.1371/journal.pone.0035645
Zebrafish chemical screening reveals the impairment of dopaminergic neuronal survival by cardiac glycosides
Abstract
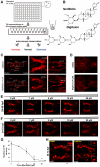
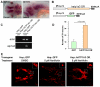
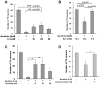
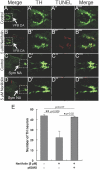
Parkinson's disease is a neurodegenerative disorder characterized by the prominent degeneration of dopaminergic (DA) neurons among other cell types. Here we report a first chemical screen of over 5,000 compounds in zebrafish, aimed at identifying small molecule modulators of DA neuron development or survival. We find that Neriifolin, a member of the cardiac glycoside family of compounds, impairs survival but not differentiation of both zebrafish and mammalian DA neurons. Cardiac glycosides are inhibitors of Na(+)/K(+) ATPase activity and widely used for treating heart disorders. Our data suggest that Neriifolin impairs DA neuronal survival by targeting the neuronal enriched Na(+)/K(+) ATPase α3 subunit (ATP1A3). Modulation of ionic homeostasis, knockdown of p53, or treatment with antioxidants protects DA neurons from Neriifolin-induced death. These results reveal a previously unknown effect of cardiac glycosides on DA neuronal survival and suggest that it is mediated through ATP1A3 inhibition, oxidative stress, and p53. They also elucidate potential approaches for counteracting the neurotoxicity of this valuable class of medications.
Conflict of interest statement
Figures

References
-
- Björklund A, Lindvall O. Dopamine-containing systems in the CNS. In: Björklund A, Hökfelt T, editors. Handbook of Chemical Neuroanatomy. Amsterdam: Elsevier; 1984. pp. 55–122.
-
- Goldstein DS, Eisenhofer G, McCarty R. Catecholamines: bridging basic science with clinical medicine; In: August JT, Anders MW, Murad F, Coyle JT, editors. San Diego, California: Academic Press; 1998.
-
- Chaudhuri KR, Schapira AH. Non-motor symptoms of Parkinson's disease: dopaminergic pathophysiology and treatment. Lancet Neurol. 2009;8:464–474. - PubMed
-
- Meissner WG, Frasier M, Gasser T, Goetz CG, Lozano A, et al. Priorities in Parkinson's disease research. Nat Rev Drug Discov. 2011;10:377–393. - PubMed
-
- Smeets WJAJ, Reiner A. Phylogeny and development of catecholamine systems in the CNS of vertebrates. Cambridge, England: Cambridge University Press; 1994.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

