Simple model of hydrophobic hydration
- PMID: 22564051
- PMCID: PMC5234790
- DOI: 10.1021/jp300743a
Simple model of hydrophobic hydration
Abstract
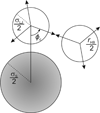
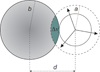
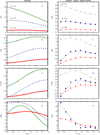
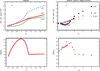
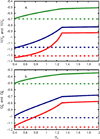
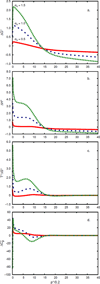
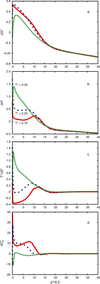
Water is an unusual liquid in its solvation properties. Here, we model the process of transferring a nonpolar solute into water. Our goal was to capture the physical balance between water's hydrogen bonding and van der Waals interactions in a model that is simple enough to be nearly analytical and not heavily computational. We develop a 2-dimensional Mercedes-Benz-like model of water with which we compute the free energy, enthalpy, entropy, and the heat capacity of transfer as a function of temperature, pressure, and solute size. As validation, we find that this model gives the same trends as Monte Carlo simulations of the underlying 2D model and gives qualitative agreement with experiments. The advantages of this model are that it gives simple insights and that computational time is negligible. It may provide a useful starting point for developing more efficient and more realistic 3D models of aqueous solvation.
Figures



References
-
- Pratt LR, Pohorille A. Chem. Rev. 2002;102:2671–2692. - PubMed
-
- Widom B, Bhimalapuram P, Koga K. Phys. Chem. Chem. Phys. 2003;5:3085–3093.
-
- Pratt LR, Chandler D. J. Chem. Phys. 1977;67:3683–3704.
-
- Ben-Naim A. Hydrophobic Interactions. New York: Plemun; 1980.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

